Question:
A solution containing $0.85 \mathrm{~g}$ of $\mathrm{ZnCl}_{2}$ in $125.0 \mathrm{~g}$ of water freezes at $-0.23^{\circ} \mathrm{C}$. The apparent degree of dissociation of the salt is :
$\left(\mathrm{k}_{f}\right.$ for water $=1.86 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$, atomic mass ; $\mathrm{Zn}=65.3$ and $\left.\mathrm{Cl}=35.5\right)$
Correct Option: , 3
Solution:
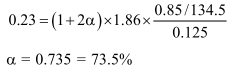
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
