Question:
A xenon compound 'A' upon partial hydrolysis gives $\mathrm{XeO}_{2} \mathrm{~F}_{2}$. The number of lone pair of electrons present in compound $A$ is_________ (Round off to the Nearest integer)
Solution:
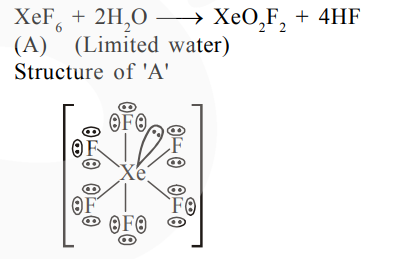
Total l.p. on $(\mathrm{A})=19$
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
