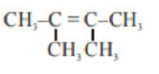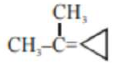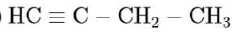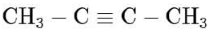An unsaturated hydrocarbon $\mathrm{X}$ on ozonolysis gives $\mathrm{A}$. Compound $\mathrm{A}$ when warmed with ammonical silver nitrate forms a bright silver mirror along the sides of the test tube. The unsaturated hydrocarbon $X$ is:
Correct Option: , 3

As (A) compound given positive tollen's test hence it may consist - $\mathrm{CHO}$ (aldehyde group). or it can be $\mathrm{HCOOH}$ So for the given option:
(c)
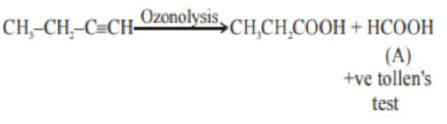
and for other compounds (options):
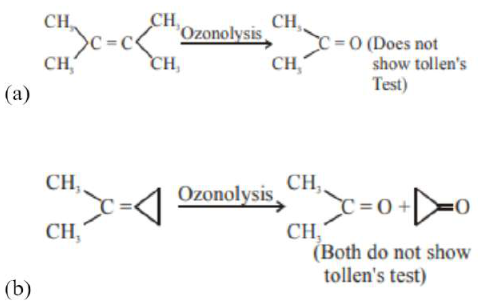
(d) $\mathrm{CH}_{3}-\mathrm{C} \equiv \mathrm{C}-\mathrm{CH}_{3} \stackrel{\text { Ozonolysis }}{\longrightarrow} 2 \mathrm{CH}_{3}-\mathrm{COOH}$
(Does not show tollen's test)
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.