Question:
Assume that a neutron breaks into a proton and an electron. The energy released during this process is:
(Mass of neutron $=1.6747 \times 10^{-27} \mathrm{~kg}$
Mass of proton $=1.6725 \times 10^{-27} \mathrm{~kg}$
Mass of electron $=9 \times 10^{-31} \mathrm{~kg}$ )
Correct Option: , 2
Solution:
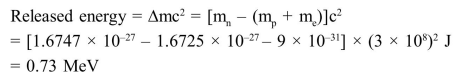
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
