Question:
In a face centred cubic lattice, atoms of A form the corner points and atoms of B form
the face centred points. If two atoms of A are missing from the corner points, the formula
of the ionic compound is
Correct Option: , 3
Solution:
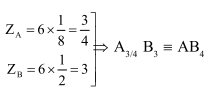
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
