Question:
Reaction of an inorganic sulphite $\mathrm{X}$ with dilute $\mathrm{H}_{2} \mathrm{SO}_{4}$ generates compound Y. Reaction of $Y$ with $\mathrm{NaOH}$ gives $X$. Further, the reaction of $\mathrm{X}$ with $\mathrm{Y}$ and water affords compound Z. Y and Z, respectively, are:
Correct Option: , 3
Solution:
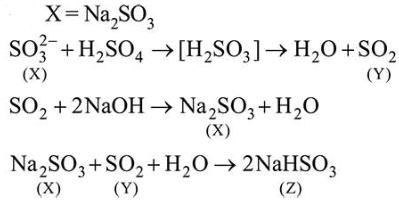
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
