Question:
Solubility product of silver bromide is $5.0 \times 10^{-13}$. The quantity of potassium bromide (molar mass taken as $120 \mathrm{~g} \mathrm{~mol}^{-1}$ ) to be added to 1 litre of $0.05 \mathrm{M}$ solution of silver nitrate to start the precipitation of $\mathrm{AgBr}$ is :-
Correct Option: , 3
Solution:
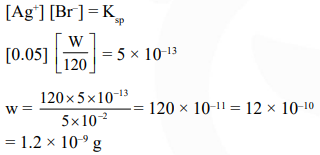
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
