Question:
The volume (in $\mathrm{mL}$ ) of $0.1 \mathrm{~N} \mathrm{NaOH}$ require to neutralise $10 \mathrm{~mL}$ of $0.1 \mathrm{~N}$ phosphinic acid is_________________ .
Solution:
(10)
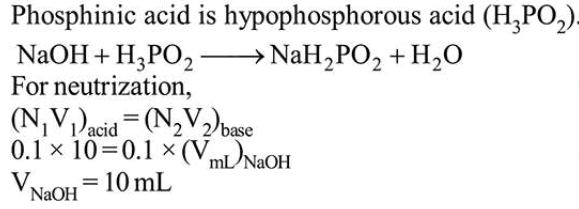
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
