The enthalpy of combustion of methane, graphite and dihydrogen at $298 \mathrm{~K}$ are, $-890.3 \mathrm{~kJ} \mathrm{~mol}^{-1}-393.5 \mathrm{~kJ} \mathrm{~mol}^{-1}$, and $-285.8 \mathrm{~kJ} \mathrm{~mol}^{-1}$ respectively. Enthalpy of formation of $\mathrm{CH}_{4(g)}$ will be
(i) $-74.8 \mathrm{~kJ} \mathrm{~mol}^{-1}$
(ii) $-52.27 \mathrm{~kJ} \mathrm{~mol}^{-1}$
(iii) $+74.8 \mathrm{~kJ} \mathrm{~mol}^{-1}$
(iv) $+52.26 \mathrm{~kJ} \mathrm{~mol}^{-1}$
According to the question,
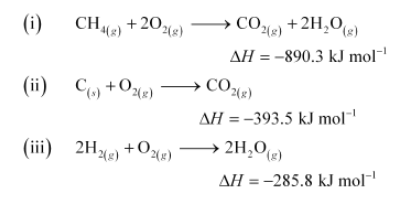
Thus, the desired equation is the one that represents the formation of CH4 (g) i.e.,
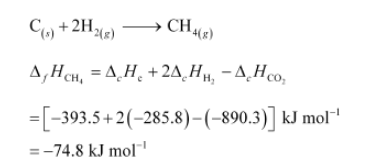
$\therefore$ Enthalpy of formation of $\mathrm{CH}_{4(g)}=-74.8 \mathrm{~kJ} \mathrm{~mol}^{-1}$
Hence, alternative (i) is correct.
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
