Question:
The intermolecular potential energy for the molecules $A$, $B, C$ and $D$ given below suggests that :
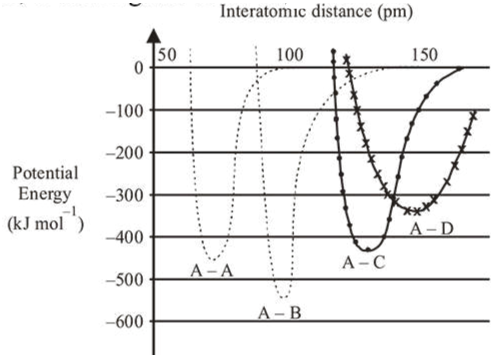
Correct Option: , 4
Solution:
A-B bond has highest intermolecular potential energy among the given molecules. Hence, it is strongest bond and has maximum bond enthalpy.
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
