The $\mathrm{Mn}^{3+}$ ion is unstable in solution and undergoes disproportionation to give $\mathrm{Mn}^{2+}, \mathrm{MnO}_{2}$, and $\mathrm{H}^{+}$ion. Write a balanced ionic equation for the reaction.
The given reaction can be represented as:
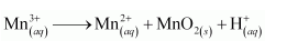
The oxidation half equation is:
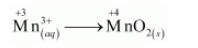
The oxidation number is balanced by adding one electron as:
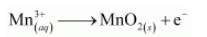
The charge is balanced by adding 4H+ ions as:
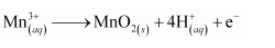
The O atoms and H+ ions are balanced by adding 2H2O molecules as:
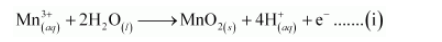
The reduction half equation is:
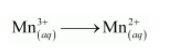
The oxidation number is balanced by adding one electron as:
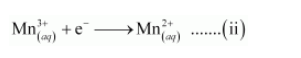
The balanced chemical equation can be obtained by adding equation (i) and (ii) as:
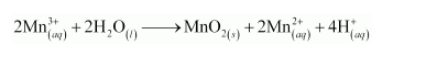
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
