Carbonyl Compound - JEE Advanced Previous Year Questions with Solutions
JEE Advanced Previous Year Questions of Chemistry with Solutions are available at eSaral. Practicing JEE Advanced Previous Year Papers Questions of Chemistry will help the JEE aspirants in realizing the question pattern as well as help in analyzing weak & strong areas.
Simulator
Previous Years JEE Advance Questions
Paragraph for Question Nos. 1 to 3
A carbonyl compound P, which gives positive iodoform test, undergoes reaction with MeMgBr followed by dehydration to give an olefin Q. Ozonolysis of Q leads to a dicarbonyl compound R, which undergoes intramolecular aldol reaction to give predominantly S.
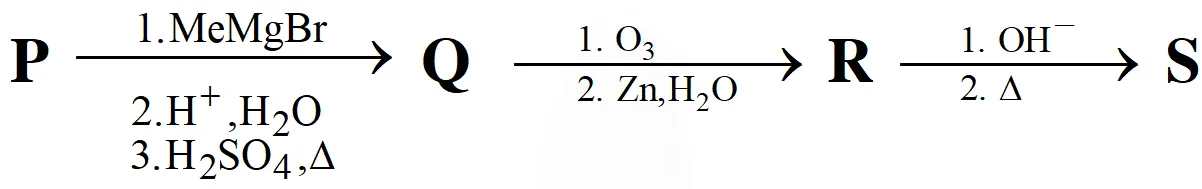 Paragraph for Questions Nos. 4 to 5
An acyclic hydrocarbon P, having molecular formula $\mathrm{C}_{6} \mathrm{H}_{10}$, gave acetone as the only organic product through the following sequence of reactions, in the which Q is an intermediate organic compound.
Paragraph for Questions Nos. 4 to 5
An acyclic hydrocarbon P, having molecular formula $\mathrm{C}_{6} \mathrm{H}_{10}$, gave acetone as the only organic product through the following sequence of reactions, in the which Q is an intermediate organic compound.
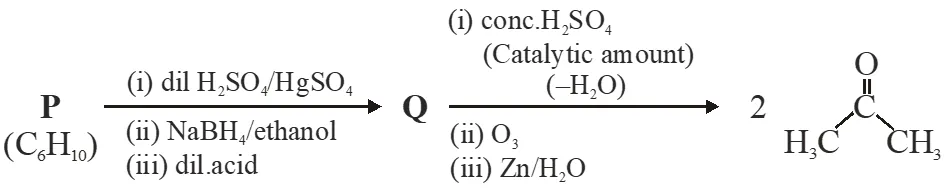
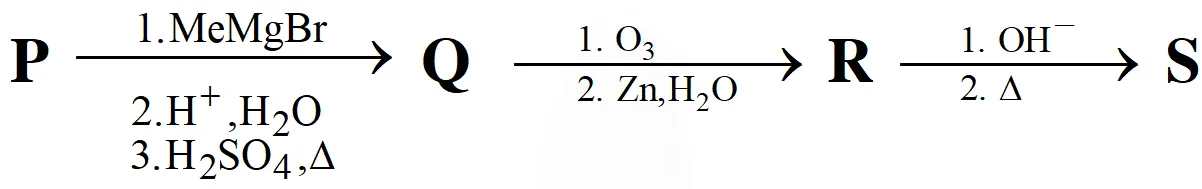
Q. The structure of the carbonyl compound P is
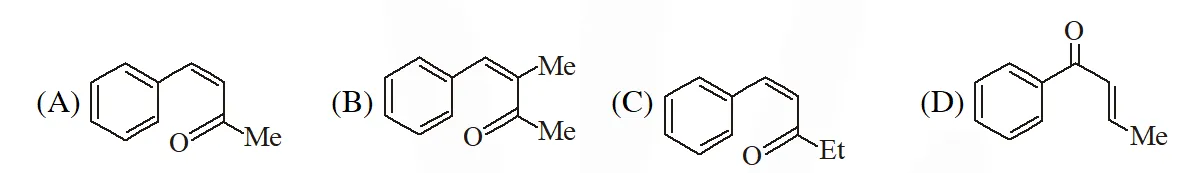 [IIT 2009]
[IIT 2009]
 [IIT 2009]
[IIT 2009]
Ans. (B)
Q. The structure of the products Q and R, respectively, are
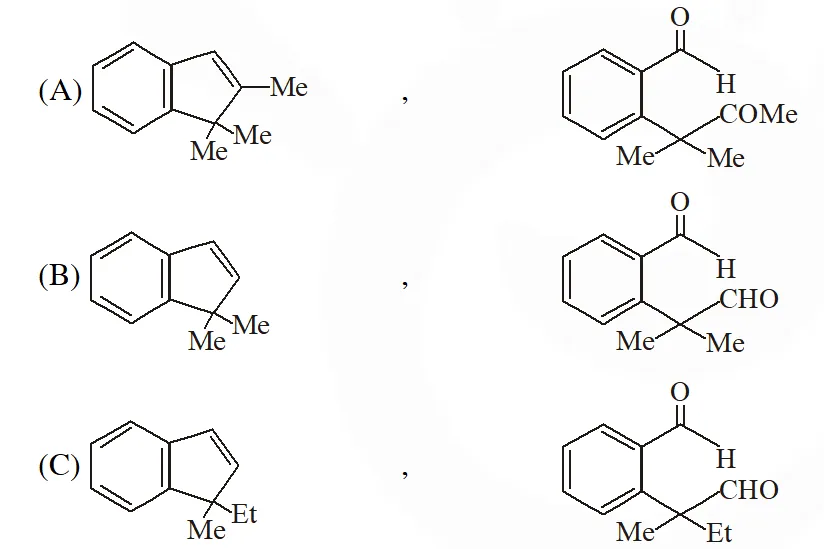
 [IIT 2009]
[IIT 2009]

 [IIT 2009]
[IIT 2009]
Ans. (A)
Q. The structure of the product S is
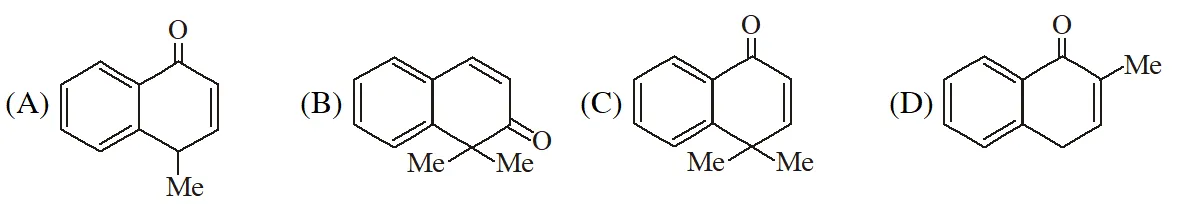 [IIT 2009]
[IIT 2009]
 [IIT 2009]
[IIT 2009]
Ans. (B)
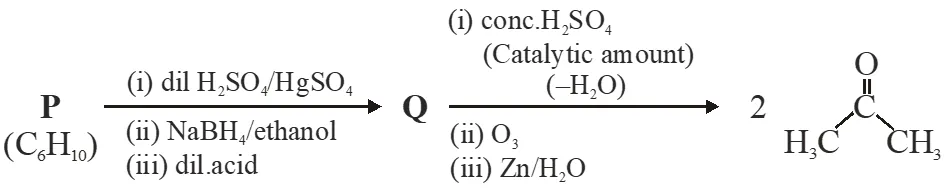
Q. The structure of compound P is -
 [IIT 2011]
[IIT 2011]
 [IIT 2011]
[IIT 2011]
Ans. (D)
Q. The structure of the compound Q is –
 [IIT 2011]
[IIT 2011]
 [IIT 2011]
[IIT 2011]
Ans. (B)
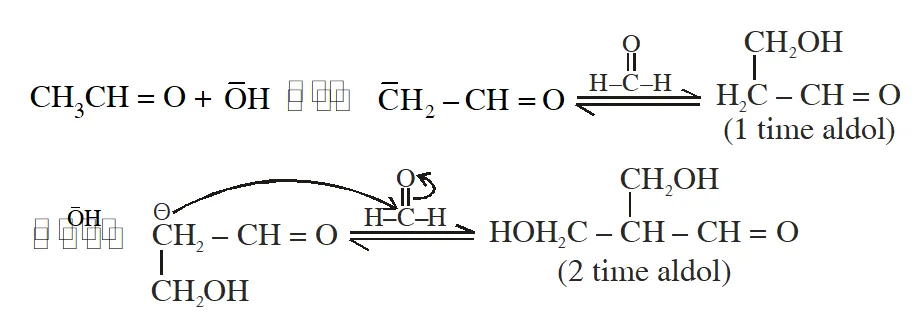
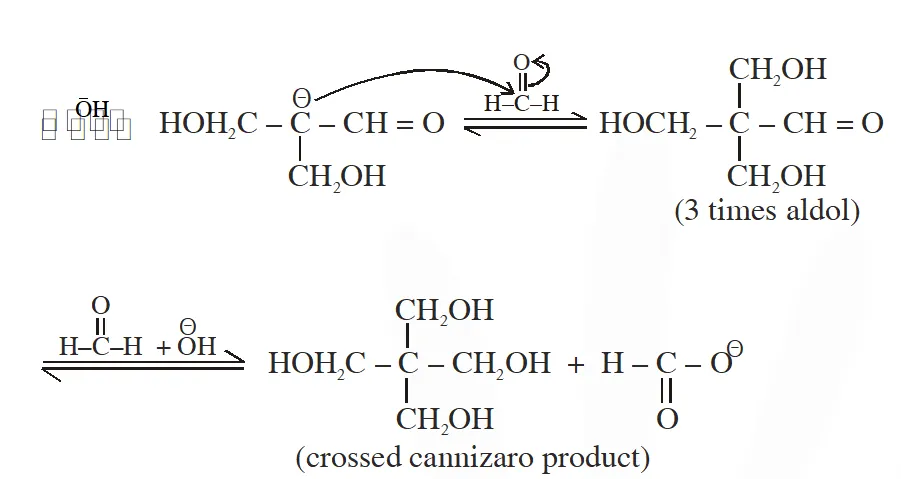
Q. The number of aldol reaction(s) that occurs in the given transformation is
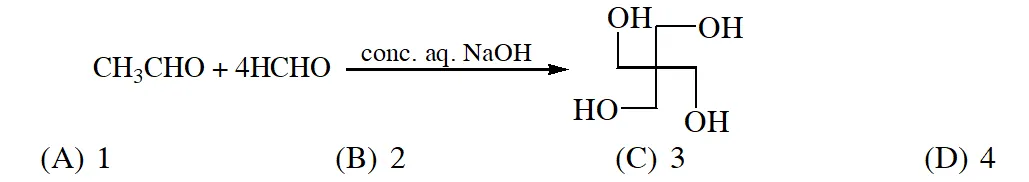 [IIT 2012]
[IIT 2012]
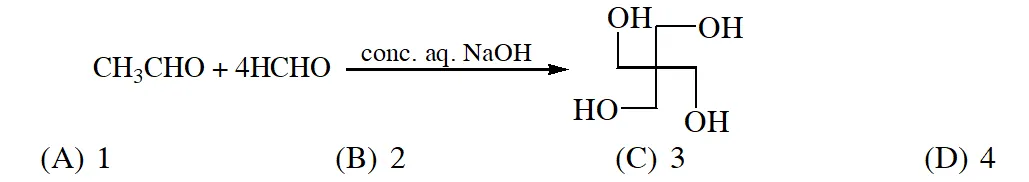 [IIT 2012]
[IIT 2012]
Ans. (C)


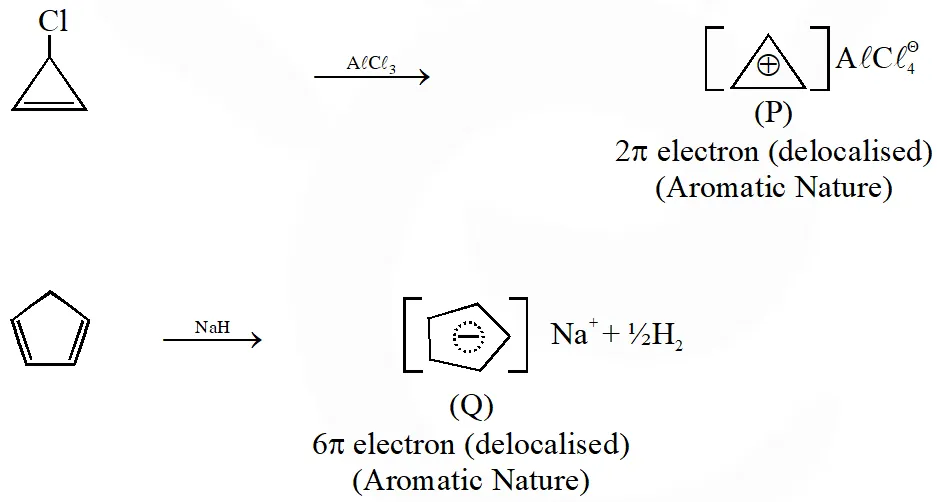
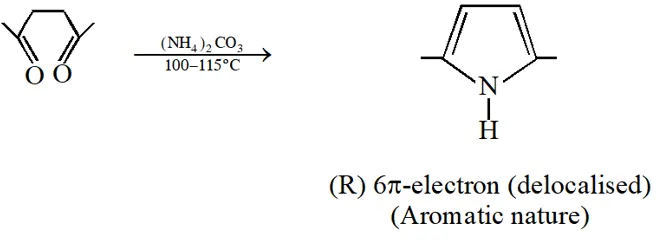
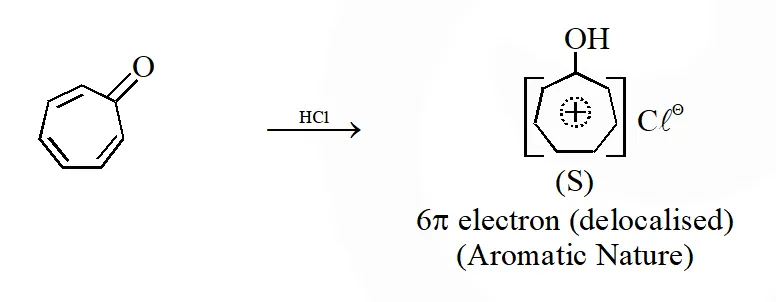
Q. Among P, Q, R and S, the aromatic compound(s) is / are :
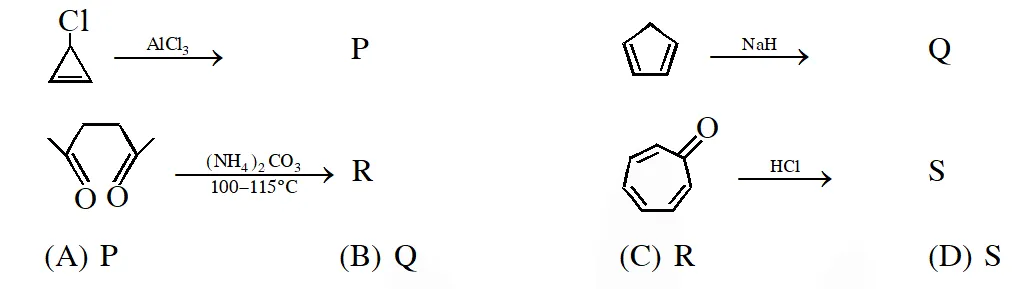 [IIT 2013]
[IIT 2013]
 [IIT 2013]
[IIT 2013]
Ans. (A,B,C,D)





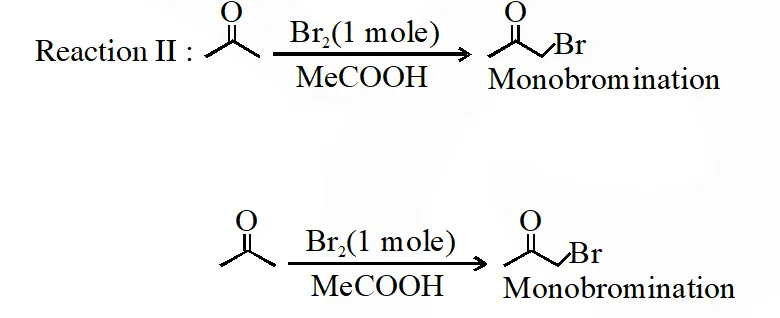
Q. After completion of the reactions (I and II), the organic compound(s) in the reaction mixtures is(are)
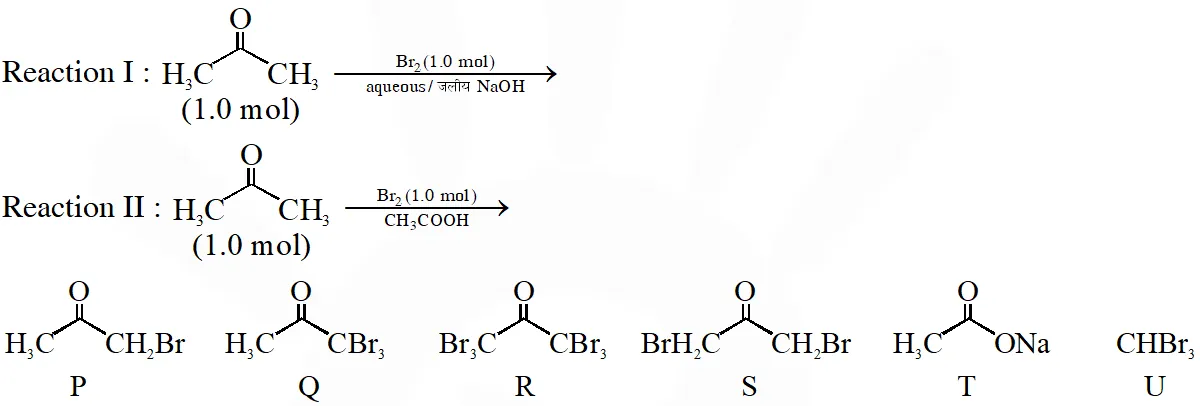 (A) Reaction I : P and Reaction II : P
(B) Reaction I : U, acetone and Reaction II : Q acetone
(C) Reaction I : T, U, acetone and Reaction II : P
(D) Reaction I : R, acetone and Reaction II : S acetone
[IIT 2013]
(A) Reaction I : P and Reaction II : P
(B) Reaction I : U, acetone and Reaction II : Q acetone
(C) Reaction I : T, U, acetone and Reaction II : P
(D) Reaction I : R, acetone and Reaction II : S acetone
[IIT 2013]
 (A) Reaction I : P and Reaction II : P
(B) Reaction I : U, acetone and Reaction II : Q acetone
(C) Reaction I : T, U, acetone and Reaction II : P
(D) Reaction I : R, acetone and Reaction II : S acetone
[IIT 2013]
(A) Reaction I : P and Reaction II : P
(B) Reaction I : U, acetone and Reaction II : Q acetone
(C) Reaction I : T, U, acetone and Reaction II : P
(D) Reaction I : R, acetone and Reaction II : S acetone
[IIT 2013]
Ans. (C)

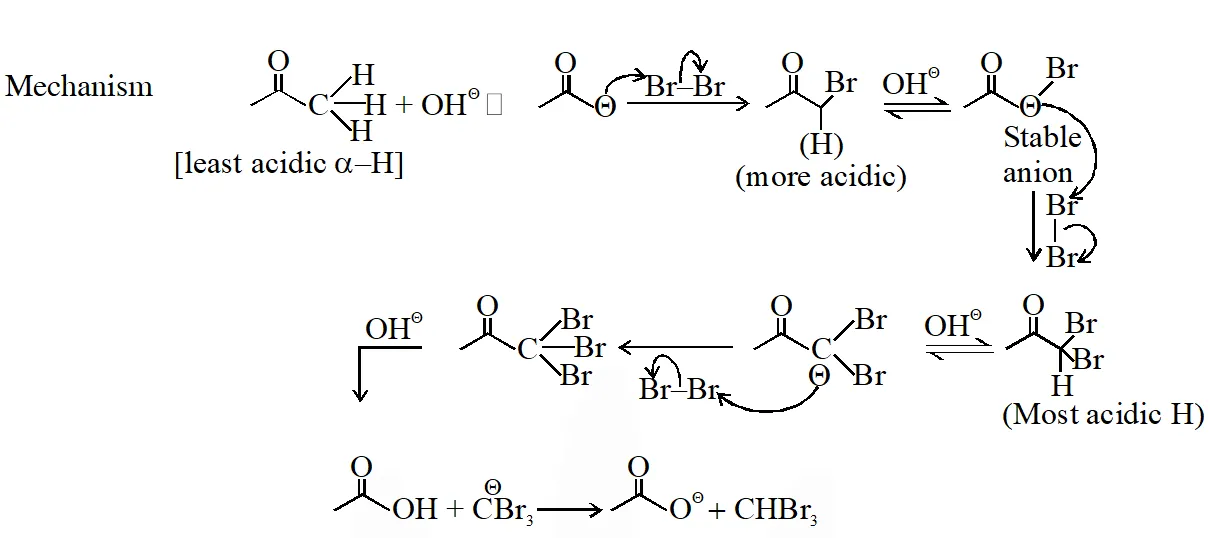 In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because -haloketones are more reactive towards base and haloform reaction takes place
In above reaction $\mathrm{Br}_{2}$ is limiting agents.
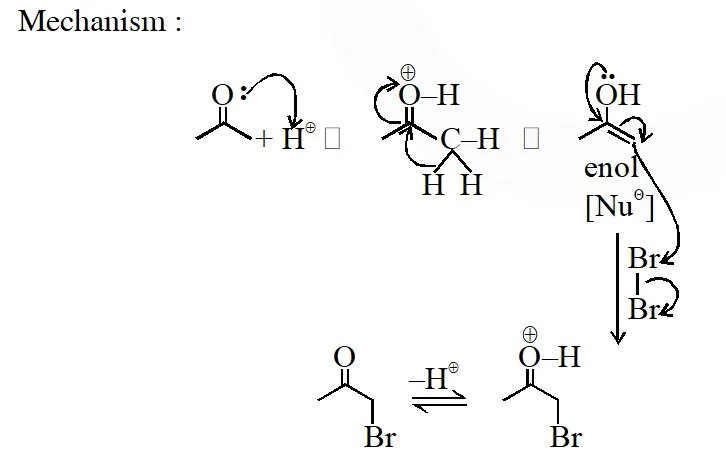
In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because -haloketones are more reactive towards base and haloform reaction takes place
In above reaction $\mathrm{Br}_{2}$ is limiting agents.

 In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because -haloketones are more reactive towards base and haloform reaction takes place
In above reaction $\mathrm{Br}_{2}$ is limiting agents.
In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because -haloketones are more reactive towards base and haloform reaction takes place
In above reaction $\mathrm{Br}_{2}$ is limiting agents.


Q. The major product in the following reaction is
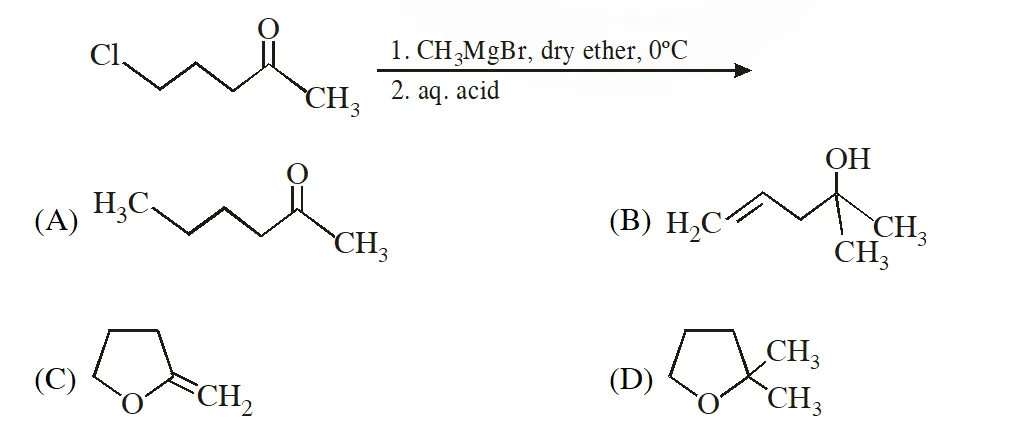 [IIT 2014]
[IIT 2014]
 [IIT 2014]
[IIT 2014]
Ans. (D)
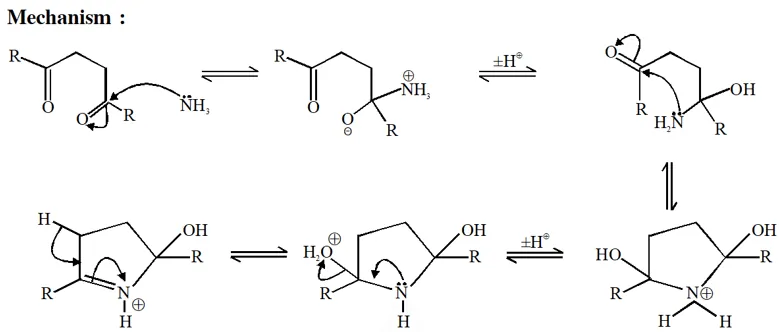
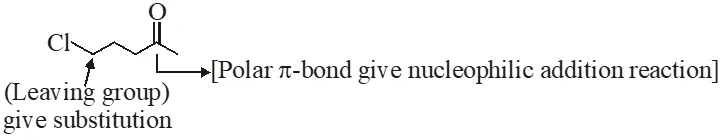 (i) Grignard prefer to give nucleophilic addition on polar $\pi$-bond and form anion intermediate.
(i) Grignard prefer to give nucleophilic addition on polar $\pi$-bond and form anion intermediate.
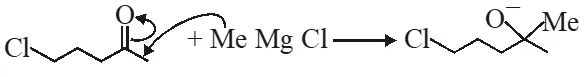 (ii) In next step anion give intramolecular nucleophilic substitution reaction & form 5 membered ring.
(ii) In next step anion give intramolecular nucleophilic substitution reaction & form 5 membered ring.
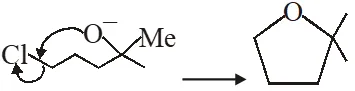
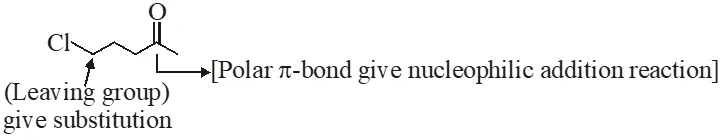 (i) Grignard prefer to give nucleophilic addition on polar $\pi$-bond and form anion intermediate.
(i) Grignard prefer to give nucleophilic addition on polar $\pi$-bond and form anion intermediate.
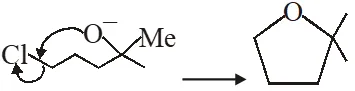
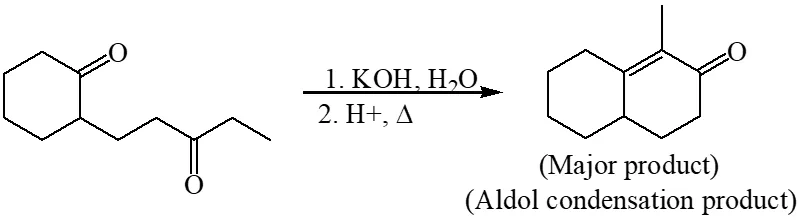
Q. The major product of the following reaction is –
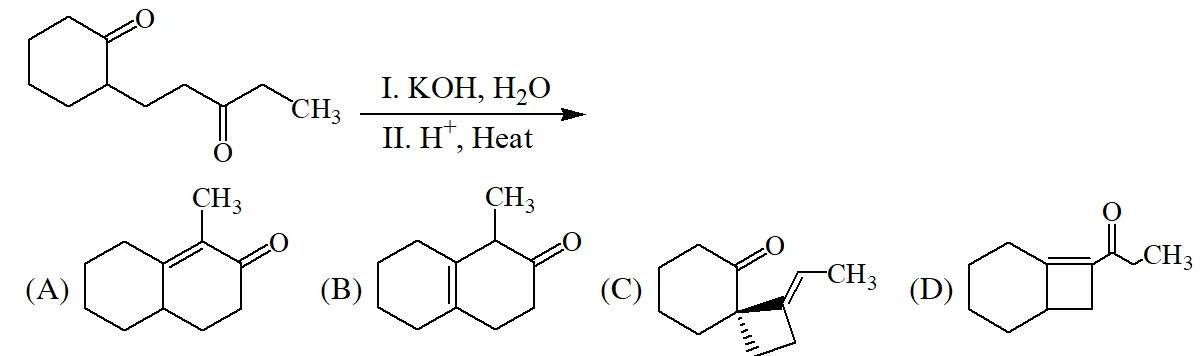 [IIT 2015]
[IIT 2015]
 [IIT 2015]
[IIT 2015]
Ans. (A)
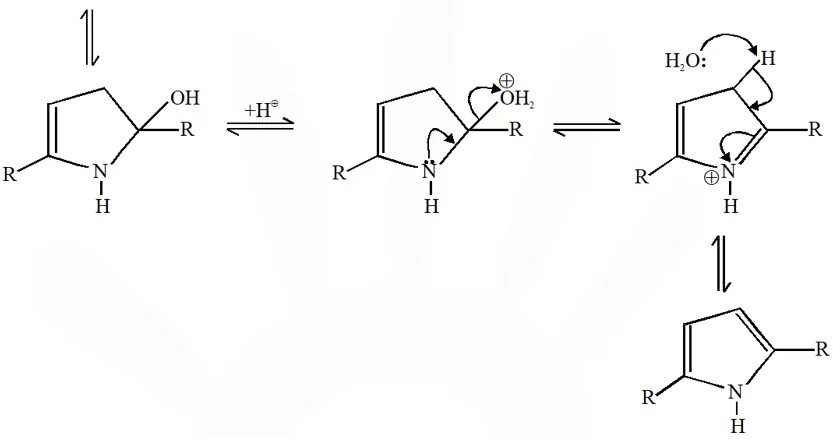
 Mechanism :
Mechanism :
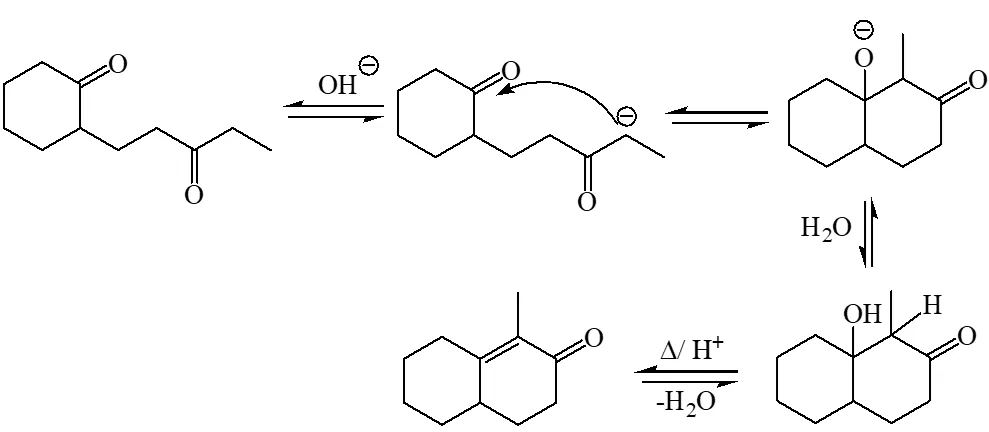
 Mechanism :
Mechanism :

Q. In the following reactions, the product S is –
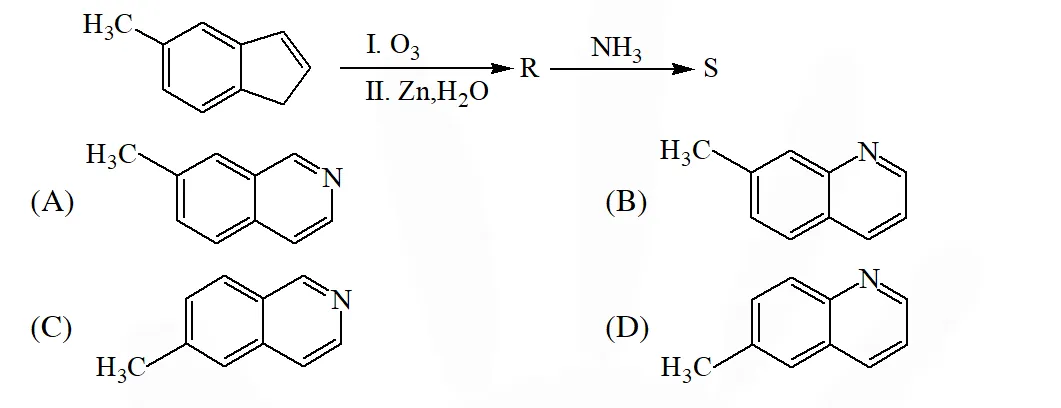 [IIT 2015]
[IIT 2015]
 [IIT 2015]
[IIT 2015]
Ans. (A)
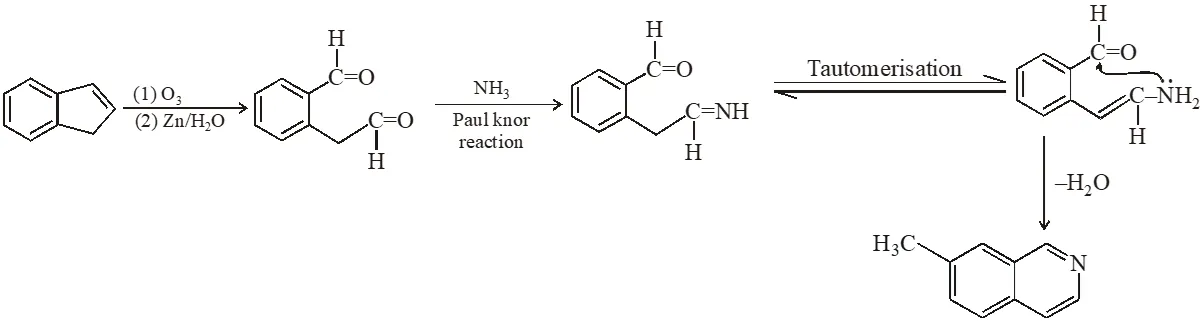

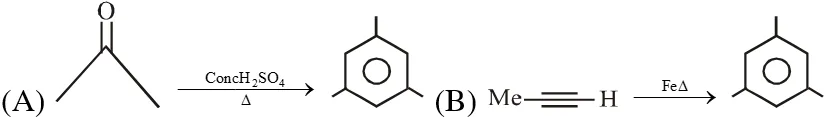
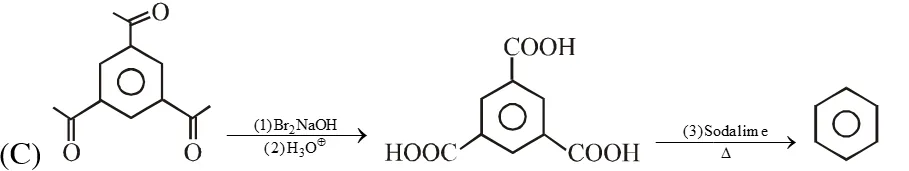
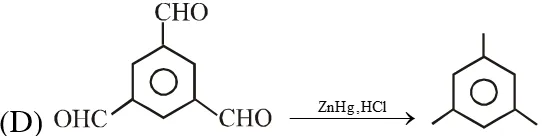
Q. The reaction(s) leading to the formation of 1,3,5-trimethylbenzene is (are)
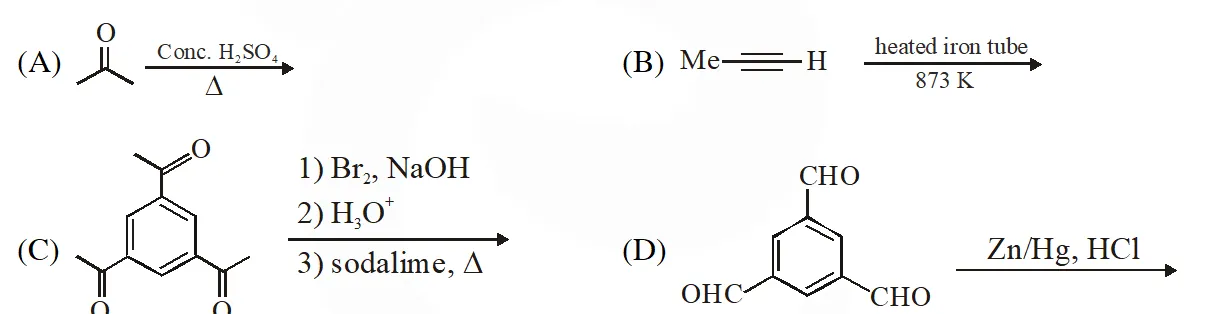 [JEE Adv. 2018]
[JEE Adv. 2018]
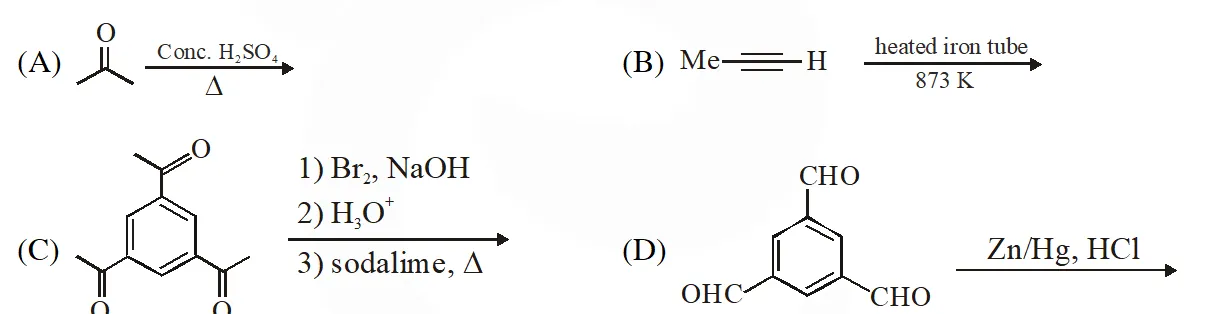 [JEE Adv. 2018]
[JEE Adv. 2018]
Ans. (A,B,D)
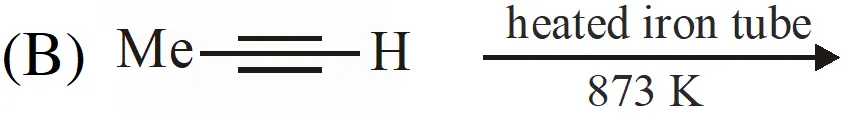



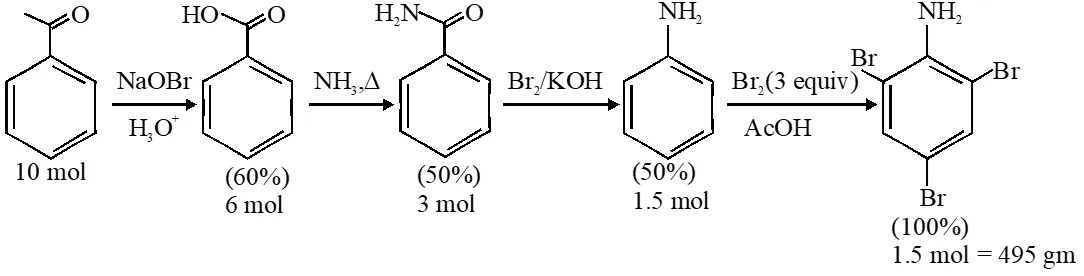
Q. In the following reaction sequence, the amount of D (in g) formed from 10 moles of acetophenone is____.
(Atomic weight in g mol–1: H = 1, C = 12, N = 14, O = 16, Br = 80. The yield (%) corresponding to the product in each step is given in the parenthesis)
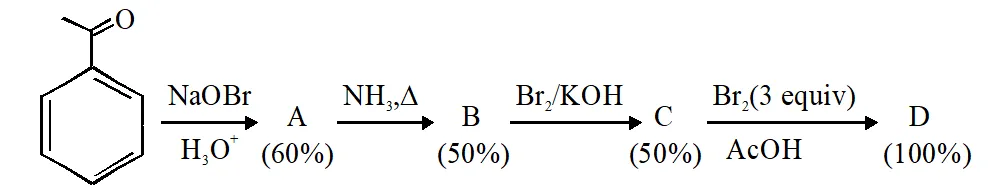 [JEE Adv. 2018]
[JEE Adv. 2018]
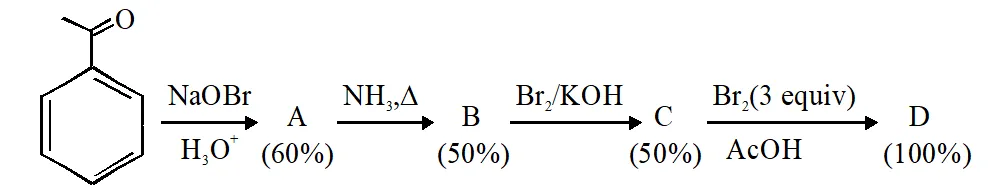 [JEE Adv. 2018]
[JEE Adv. 2018]
Ans. 495