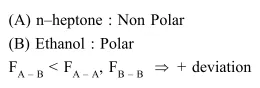A binary liquid solution is prepared by mixing n-heptane and ethanol.
Question:
A binary liquid solution is prepared by mixing n-heptane and ethanol. Which one of the folloowing
statements is correct regarding the behaviour of the solution ?
Correct Option: , 4
Solution: