A compound C (molecular formula, C2H4O2) reacts
Question:
A compound C (molecular formula, C2H4O2) reacts with Na-metal to form a compound R and evolves a gas which burns with a pop sound. Compound
C on treatment with an alcohol A in presence of an acid forms a sweet smelling compound S (molecular formula, C3H6O2). On addition of NaOH to C,
it also gives R and water. S on treatment with NaOH solution gives back R and A.
Identify C, R, A, S and write down the reactions involved.
Solution:
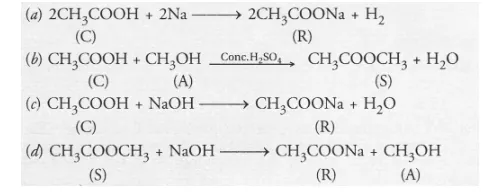
From the available information, it is evident that
- Compound C with molecular formula C2H4O2 is ethanoic acid (CH3COOH)
- The compound R is sodium ethanoate and has formula CH3COONa
- Since the compound S has only three carbon atoms (C3H6O2) and has been formed by the action of an alcohol on compound C (C2H4O2), this means that the alcohol A has only one carbon atom. It is methanol (CH3OH).
- The compound S with a sweet smell is methyl ethanoate with formula CH3COOCH3.
The chemical reactions involved are as follows :