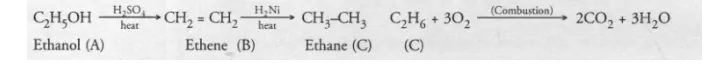Question:
An organic compound A on heating with concentrated H2SO4 forms a compound B which on addition of one mole of hydrogen in presence of Ni forms a
compound C. One mole of compound C on combustion forms two moles of CO2 and three moles of H2O. Identify the compounds A, B and C and write
the chemical equations for the reactions involved.
Solution:
Since one mole of compound C on combustion forms two moles of CO2 and three moles of H2O the compound C is a hydrocarbon with formula
C2H6. It is ethane. The compound B which forms C2H6 upon addition of hydrogen is ethene (C2H4). The organic compound A which forms ethene
upon acidic dehydration is ethanol. The chemical equations for the reactions involved are :