Arrange the following conformational isomers of n-butane in order of their increasing potential energy:
Question:
Arrange the following conformational isomers of n-butane in order of their increasing potential energy:
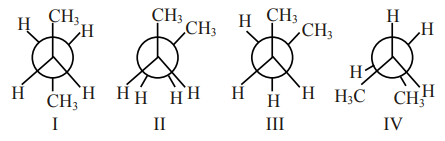
Correct Option: , 4
Solution:
More stable less potential energy.
Stability order : I > III > IV > II
So
Potential energy: II $>$ IV $>$ III $>$ I
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
