Question: Consider an ideal gas with the following distribution of speeds
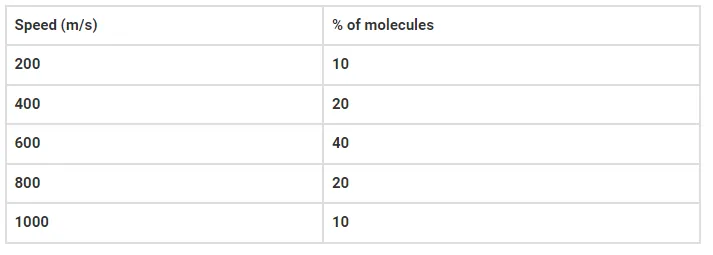
(a) calculate Vrms and hence T (m = 3.0 × 10-26 kg)
(b) if all the molecules with speed 1000 m/s escape from the system, calculate new Vrms and hence T
Solution:
(a) T = 296 K
(b) T = 248.04 K