Question:
How would you bring about the following conversions ? Name the process and write the reaction involved.
(a) ethanol to ethene.
(b) Methanol to Ethanoic acid. Write the reactions.
Solution:
(a) From ethanol: This method involves the slow oxidation of a dilute solution of ethanol (10-15 per cent) by oxygen present in air in the presence of an enzyme acetobactor.
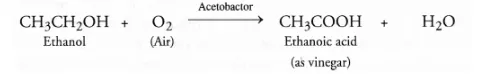
The acid obtained is in the form of dilute solution called vinegar.
We have also studied under ethanol that it gets oxidised to ethanoic acid in the presence of dilute solution of alkaline KMnO4 or acidified K2Cr2O7.
(b) From methanol: These days ethanoic acid is manufactured by the reaction between methanol and carbon monoxide in the presence of iodine-
rhodium (I2 — Rh) catalyst mixture.

