Question:
lood reducing nature of $\mathrm{H}_{3} \mathrm{PO}_{2}$ ttributed to the presence of:
Correct Option: , 3
Solution:
$\mathrm{H}_{3} \mathrm{PO}_{2}$ is good reducing agent due to presence
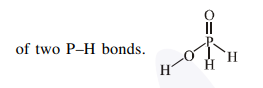
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
