Question:
Predict which of the following will be coloured in aqueous solution? Ti3+, V3+, Cu+, Sc3+, Mn2+, Fe3+ and Co2+. Give reasons for each.
Solution:
Only the ions that have electrons in d-orbital and in which d-d transition is possible will be coloured. The ions in which d-orbitals are empty or completely filled will be colourless as no d-d transition is possible in those configurations.
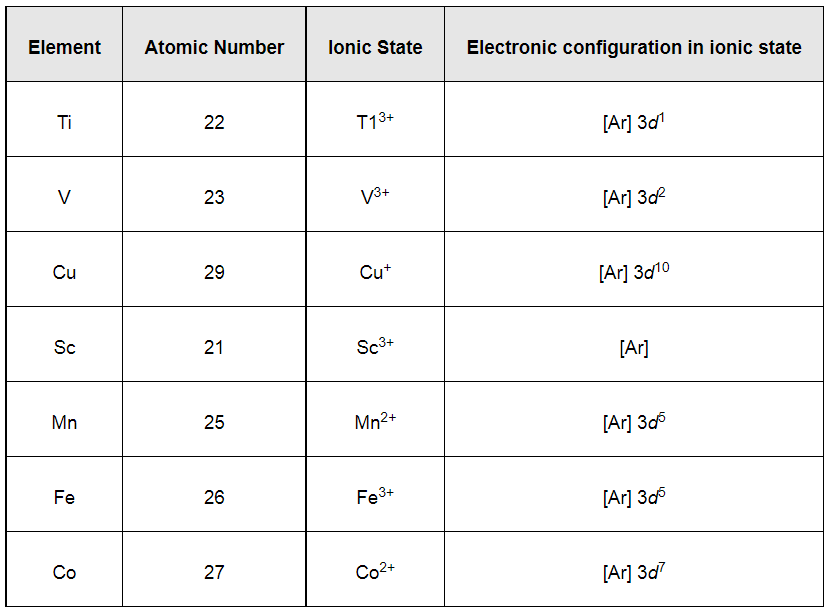
From the above table, it can be easily observed that only Sc3+ has an empty d-orbital and Cu+ has completely filled d-orbitals. All other ions, except Sc3+ and Cu+, will be coloured in aqueous solution because of d−d transitions.
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
