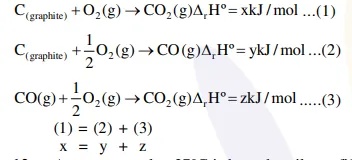Question:
Given :
(i) $\mathrm{C}$ (graphite) $+\mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{CO}_{2}(\mathrm{~g})$;
$\Delta \mathrm{rH}^{\circ}=\mathrm{x} \mathrm{kJ} \mathrm{mol}^{-1}$
(ii) $\mathrm{C}$ (graphite) $+\frac{1}{2} \mathrm{O}_{2}$ (g) $\rightarrow \mathrm{CO}_{2}(\mathrm{~g})$;
$\Delta \mathrm{rH}^{\circ}=\mathrm{y} \mathrm{kJ} \mathrm{mol}{ }^{-1}$
(iii) $\mathrm{CO}(\mathrm{g})+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{CO}_{2}(\mathrm{~g})$;
$\Delta \mathrm{rH}^{\circ}=\mathrm{z} \mathrm{kJ} \mathrm{mol}-1$
Based on the above thermochemical equations, find out which one of the following algebraic relationships is correct?
Correct Option: , 3
Solution: