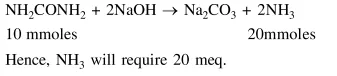Question: The ammonia $\left(\mathrm{NH}_{3}\right)$ released on quantitative reaction of $0.6 \mathrm{~g}$ urea $\left(\mathrm{NH}_{2} \mathrm{CONH}_{2}\right)$ with sodium hydroxide $(\mathrm{NaOH})$ can be neutralized by:
$100 \mathrm{ml}$ of $0.1 \mathrm{~N} \mathrm{HCl}$
$200 \mathrm{ml}$ of $0.4 \mathrm{~N} \mathrm{HCl}$
$100 \mathrm{ml}$ of $0.2 \mathrm{~N} \mathrm{HCl}$
$200 \mathrm{ml}$ of $0.2 \mathrm{~N} \mathrm{HCl}$
Correct Option: , 3
Solution: