The above reaction requires which of the following
Question:
The above reaction requires which of the following reaction conditions?
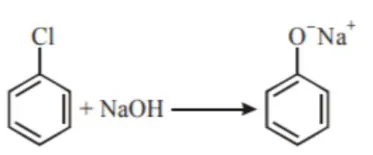
Correct Option: , 4
Solution:
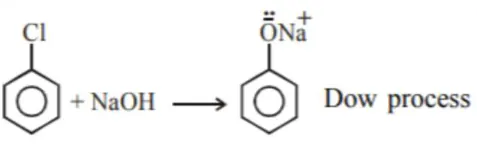
Temperature $=623 \mathrm{~K}$
Pressure $=300 \mathrm{~atm}$
