The concentration of sulphide ion in $0.1 \mathrm{M} \mathrm{HCl}$ solution saturated with hydrogen sulphide is $1.0 \times 10^{-19} \mathrm{M}$. If $10 \mathrm{~mL}$ of this is added to $5 \mathrm{~mL}$ of $0.04 \mathrm{M}$ solution of the following: $\mathrm{FeSO}_{4}, \mathrm{MnCl}_{2}, \mathrm{ZnCl}_{2}$ and $\mathrm{CdCl}_{2}$. in which of these solutions precipitation will take place?
Given $K_{s p}$ for FeS $=6.3 \times 10^{-18}, \mathrm{MnS}=2.5 \times 10^{-13}, \mathrm{ZnS}=1.6 \times 10^{-24}$,
$\mathrm{CdS}=8.0 \times 10^{-27}$
For precipitation to take place, it is required that the calculated ionic product exceeds the Ksp value.
Before mixing:
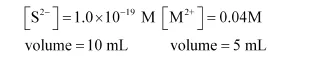
After mixing:
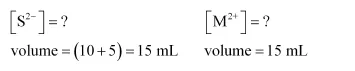
$\left[\mathrm{S}^{2-}\right]=\frac{1.0 \times 10^{-19} \times 10}{15}=6.67 \times 10^{-20} \mathrm{M}$
$\left[\mathrm{M}^{2+}\right]=\frac{0.04 \times 5}{15}=1.33 \times 10^{-2} \mathrm{M}$
Ionic product $=\left[\mathrm{M}^{2+}\right]\left[\mathrm{S}^{2-}\right]$
$=\left(1.33 \times 10^{-2}\right)\left(6.67 \times 10^{-20}\right)$
$=8.87 \times 10^{-22}$
This ionic product exceeds the $K_{s p}$ of $Z n s$ and $C d S$. Therefore, precipitation will occur in $\mathrm{CdCl}_{2}$ and $\mathrm{ZnCl}_{2}$ solutions.
