Question: The correct statement among the following is
$\left(\mathrm{SiH}_{3}\right)_{3} \mathrm{~N}$ is pyramidal and more basic than $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
$\left(\mathrm{SiH}_{3}\right)_{3} \mathrm{~N}$ is planar and more basic than $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
$\left(\mathrm{SiH}_{3}\right)_{3} \mathrm{~N}$ is pyramidal and less basic than $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
$\left(\mathrm{SiH}_{3}\right)_{3} \mathrm{~N}$ is planar and less basic than $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
Correct Option: , 4
Solution:
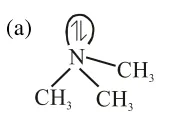
nitrogen is $\mathrm{sp}^{3}$ hybrid and pyramidal no back-bonding i.e.more basic
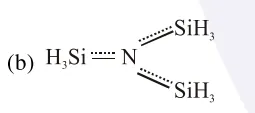
Nitrogen $\mathrm{sp}^{2}$ hybrid and planar due to back bonding and less basic because lone pair is not available for donation.


