Question: The decreasing order of basicity of the following amines is:
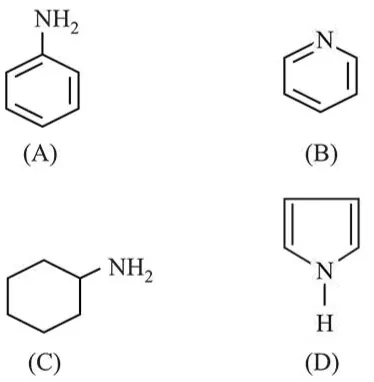
$(\mathrm{A})>(\mathrm{C})>(\mathrm{D})>(\mathrm{B})$
$(\mathrm{C})>(\mathrm{A})>(\mathrm{B})>(\mathrm{D})$
$(\mathrm{B})>(\mathrm{C})>(\mathrm{D})>(\mathrm{A})$
$(\mathrm{C})>(\mathrm{B})>(\mathrm{A})>(\mathrm{D})$
Correct Option: , 4
Solution:
Basic strength of amines depends upon availability of lone pair of electrons. Aliphatic amines are more basic than aromatic amines.