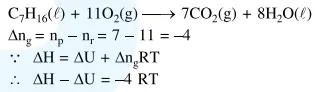The difference between
Question:
The difference between $\Delta \mathrm{H}$ and $\Delta \mathrm{U}(\Delta \mathrm{H}-\Delta \mathrm{U})$, when the combustion of one mole of heptane (1) is carried out at a temperature $T$, is equal to:
Correct Option: , 3
Solution: