The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen.
The drain cleaner, Drainex contains small bits of aluminum which react with caustic soda to produce dihydrogen. What volume of dihydrogen at 20 °C and one bar will be released when 0.15g of aluminum reacts?
The reaction of aluminium with caustic soda can be represented as:
$2 \mathrm{Al}+2 \mathrm{NaOH}+2 \mathrm{H}_{2} \mathrm{O} \longrightarrow 2 \mathrm{NaAlO}_{2}+3 \mathrm{H}_{2}$
$2 \times 27 \mathrm{~g}$
$3 \times 22400 \mathrm{~mL}$
At STP (273.15 K and 1 atm), 54 g (2 × 27 g) of Al gives 3 × 22400 mL of H2..
$\therefore 0.15 \mathrm{~g}$ Al gives $\frac{3 \times 22400 \times 0.15}{54} \mathrm{~mL}$ of $\mathrm{H}_{2}$ i.e., $186.67 \mathrm{~mL}$ of $\mathrm{H}_{2}$
At STP,
$p_{1}=1 \mathrm{~atm}$
$V_{1}=186.67 \mathrm{~mL}$
$T_{1}=273.15 \mathrm{~K}$
Let the volume of dihydrogen be![]() at p2 = 0.987 atm (since 1 bar = 0.987 atm) and T2 = 20°C = (273.15 + 20) K = 293.15 K..
at p2 = 0.987 atm (since 1 bar = 0.987 atm) and T2 = 20°C = (273.15 + 20) K = 293.15 K..
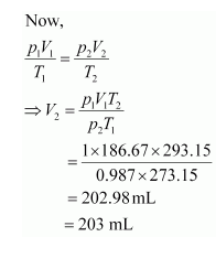
Therefore, 203 mL of dihydrogen will be released.
Click here to get exam-ready with eSaral
For making your preparation journey smoother of JEE, NEET and Class 8 to 10, grab our app now.
