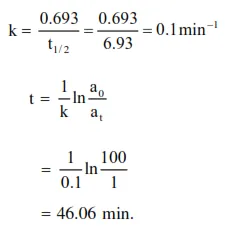Question: The half life period of a first order chemical reaction is $6.93$ minutes. The time required for the completion of $99 \%$ of the chemical reaction will be $(\log 2=0.301):-$
$46.06$ minutes
$460.6$ minutes
$230.3$ minutes
$23.03$ minutes
Correct Option: 1
Solution: