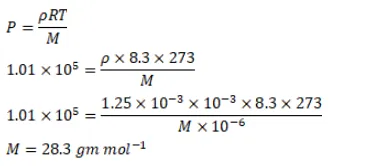Use R=8.3 J/mol-K wherever required.
Question:
Use $\mathrm{R}=8.3 \mathrm{~J} / \mathrm{mol}-\mathrm{K}$ wherever required.
The density of an ideal gas is $1.25 \times 10^{-3} \mathrm{~g} / \mathrm{cm}^{3}$ at STP. Calculate the molecular weight of the gas.
Solution: