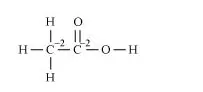What are the oxidation numbers of the underlined elements in each of the following and how do you rationalise your results?
What are the oxidation numbers of the underlined elements in each of the following and how do you rationalise your results?
(a) $\mathrm{K} \underline{\mathrm{I}}_{3}$
(b) $\mathrm{H}_{2} \mathrm{~S}_{4} \mathrm{O}_{6}$
(c) $\mathrm{Fe}_{3} \mathrm{O}_{4}$
(d) $\underline{\mathrm{C}} \mathrm{H}_{3} \underline{\mathrm{C}} \mathrm{H}_{2} \mathrm{OH}$
(e) $\underline{\mathrm{C}} \mathrm{H}_{3} \underline{\mathrm{C}} \mathrm{O} \mathrm{OH}$
(a) KI3
In $\mathrm{KI}_{3}$, the oxidation number ( $\mathrm{O} . \mathrm{N}$.) of $\mathrm{K}$ is $+1$. Hence, the average oxidation number of $\mathrm{I}$ is $-\frac{1}{3}$. However, $\mathrm{O} . \mathrm{N}$. cannot be fractional. Therefore, we will have to consider the structure of $\mathrm{Kl}_{3}$ to find the oxidation states.
In a $\mathrm{Kl}_{3}$ molecule, an atom of iodine forms a coordinate covalent bond with an iodine molecule.
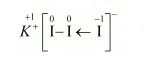
Hence, in a KI3molecule, the O.N. of the two I atoms forming the I2molecule is 0, whereas the O.N. of the I atom forming the coordinate bond is –1.
(b) $\mathrm{H}_{2} \mathrm{~S}_{4} \mathrm{O}_{6}$
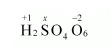
Now, $2(+1)+4(x)+6(-2)=0$
$\Rightarrow 2+4 x-12=0$
$\Rightarrow 4 x=10$
$\Rightarrow x=+2 \frac{1}{2}$
However, O.N. cannot be fractional. Hence, S must be present in different oxidation states in the molecule.
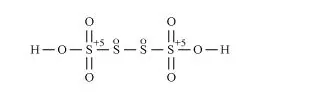
The O.N. of two of the four $S$ atoms is $+5$ and the $O . N$. of the other two $S$ atoms is 0 .
(c) $\mathrm{Fe}_{3} \mathrm{O}_{4}$
On taking the O.N. of $\mathrm{O}$ as $-2$, the O.N. of Fe is found to be $+2 \frac{2}{3}$. However, O.N. cannot be fractional.
Here, one of the three Fe atoms exhibits the O.N. of $+2$ and the other two Fe atoms exhibit the O.N. of $+3$.
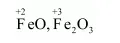
(d) $\mathrm{CH}_{3} \underline{\mathrm{CH}}_{2} \mathrm{OH}$
$2(x)+6(+1)+1(-2)=0$
or, $2 x+4=0$
or, $x=-2$
Hence, the $O \cdot N$. of $C$ is $-2$.
(e) $\underline{\mathrm{CH}}_{3} \underline{\mathrm{COOH}}$
$2(x)+4(+1)+2(-2)=0$
or, $2 x=0$
or, $x=0$
However, 0 is average O.N. of C. The two carbon atoms present in this molecule are present in different environments. Hence, they cannot have the same oxidation number. Thus, $\mathrm{C}$ exhibits the oxidation states of $+2$ and $-2$ in $\mathrm{CH}_{3} \mathrm{COOH}$.