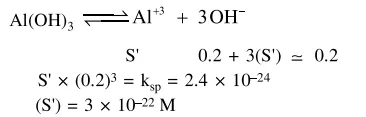What is the molar solubility of
Question:
What is the molar solubility of $\mathrm{Al}(\mathrm{OH})_{3}$ in $0.2 \mathrm{M} \mathrm{NaOH}$ solution ? Given that, solubility product of $\mathrm{Al}(\mathrm{OH})_{3}=2.4 \times 10^{-24}$ :
Correct Option: , 4
Solution: