What will be the pressure of the gaseous mixture when 0.5 L of H2 at 0.8 bar and 2.0 L of dioxygen at 0.7 bar are introduced in a 1L vessel at 27°C?
Let the partial pressure of H2 in the vessel be![]() .
.
Now,
$\begin{array}{ll}p_{1}=0.8 \text { bar } & p_{2}=p_{H_{2}}=? \\ V_{1}=0.5 \mathrm{~L} & V_{2}=1 \mathrm{~L}\end{array}$
It is known that,
$p_{1} V_{1}=p_{2} V_{2}$
$\Rightarrow p_{2}=\frac{p_{1} V_{1}}{V_{2}}$
$\Rightarrow p_{H_{2}}=\frac{0.8 \times 0.5}{1}$
$=0.4 \mathrm{bar}$
Now, let the partial pressure of $\mathrm{O}_{2}$ in the vessel be $p_{\mathrm{O}_{2}}$.
Now,
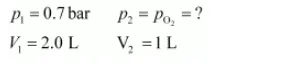
$\mathrm{p}_{1} \mathrm{~V}_{1}=\mathrm{p}_{2} \mathrm{~V}_{2}$
$\Rightarrow \mathrm{p}_{2}=\frac{\mathrm{p}_{1} \mathrm{v}_{1}}{\mathrm{v}_{2}}$
$\Rightarrow \mathrm{PO}_{2}=\frac{0.7 \times 2.0}{1}=1.4 \mathrm{bar}$
Total pressure of the gas mixture in the vessel can be obtained as:
$p_{\text {total }}=p_{\mathrm{H}_{2}}+p_{\mathrm{O}_{2}}$
$=0.4+1.4$
$=1.8$ bar
Hence, the total pressure of the gaseous mixture in the vessel is $1.8$ bar .
