Which one of the following graphs between molar conductivity
Question:
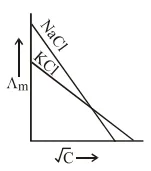
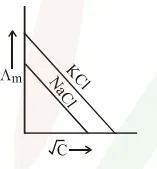
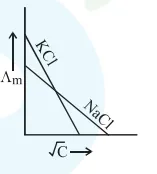
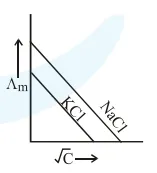
Which one of the following graphs between molar conductivity $\left(\Lambda_{\mathrm{m}}\right)$ versus $\sqrt{\mathrm{C}}$ is correct?
Correct Option: , 2
Solution:
Both $\mathrm{NaCl}$ and $\mathrm{KCl}$ are strong electrolytes and as $\mathrm{Na}^{+}$(aq.) has less conductance than $\mathrm{K}^{+}$(aq.) due to more hydration therefore the graph of option (2) is correct.