Why is Wurtz reaction not preferred for the preparation of alkanes containing odd number of carbon atoms?
Why is Wurtz reaction not preferred for the preparation of alkanes containing odd number of carbon atoms? Illustrate your answer by taking one example.
Wurtz reaction is limited for the synthesis of symmetrical alkanes (alkanes with an even number of carbon atoms) In the reaction, two similar alkyl halides are taken as reactants and an alkane, containing double the number of carbon atoms, are formed. Example:
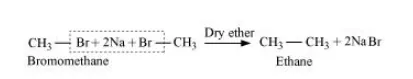
Wurtz reaction cannot be used for the preparation of unsymmetrical alkanes because if two dissimilar alkyl halides are taken as the reactants, then a mixture of alkanes is obtained as the products. Since the reaction involves free radical species, a side reaction also occurs to produce an alkene. For example, the reaction of bromomethane and iodoethane gives a mixture of alkanes.
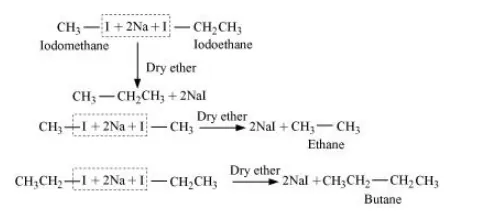
The boiling points of alkanes (obtained in the mixture) are very close. Hence, it becomes difficult to separate them
