Hydrocarbon - JEE Advanced Previous Year Questions with Solutions
JEE Advanced Previous Year Questions of Chemistry with Solutions are available at eSaral. Practicing JEE Advanced Previous Year Papers Questions of Chemistry will help the JEE aspirants in realizing the question pattern as well as help in analyzing weak & strong areas.
Simulator
Previous Years JEE Advance Questions
Paragraph For Question 3 and 4
Schemes 1 and 2 describe sequential transformation of alkynes M and N. Consider only the major products formed in each step for both the schemes.
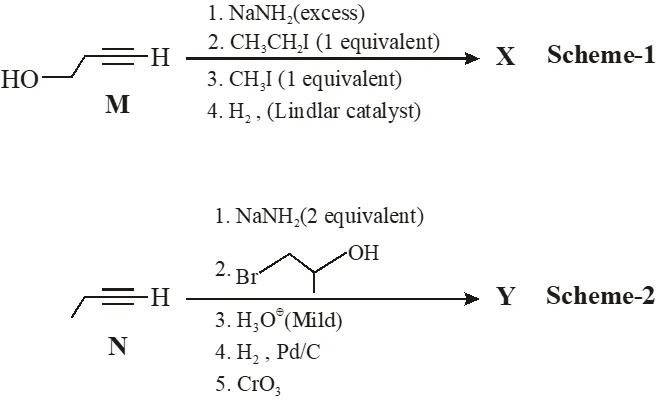 Paragraph For Questions 6 and 7
In the following reaction
Paragraph For Questions 6 and 7
In the following reaction
Q. The synthesis of 3-octyne is achieved by adding a bromoalkane into a mixture of sodium amide and an alkyne. The bromoalkane and alkyne respectively are
(A) $\mathrm{BrCH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{3}$ and $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{C} \equiv \mathrm{CH}$
(B) $\mathrm{BrCH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{3}$ and $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{C} \equiv \mathrm{CH}$
(C) $\mathrm{BrCH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{3}$ and $\mathrm{CH}_{3} \mathrm{C} \equiv \mathrm{CH}$
(D) $\mathrm{BrCH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{3}$ and $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{C} \equiv \mathrm{CH}$
[IIT-2010]
Ans. (D)
Q. Isomers of hexane, based on their branching, can be divided into three distinct classes as shown in the figure.
 The correct order of their boiling point is
(A) I > II > III
(B) III > II > I
(C) II > III > I
(D) III > I > II
[IIT-2014]
The correct order of their boiling point is
(A) I > II > III
(B) III > II > I
(C) II > III > I
(D) III > I > II
[IIT-2014]
 The correct order of their boiling point is
(A) I > II > III
(B) III > II > I
(C) II > III > I
(D) III > I > II
[IIT-2014]
The correct order of their boiling point is
(A) I > II > III
(B) III > II > I
(C) II > III > I
(D) III > I > II
[IIT-2014]
Ans. (B)

Q. The product X is -
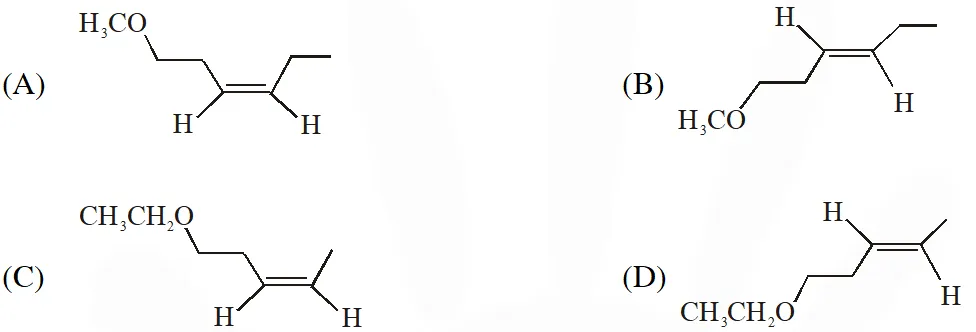 [IIT-2014]
[IIT-2014]
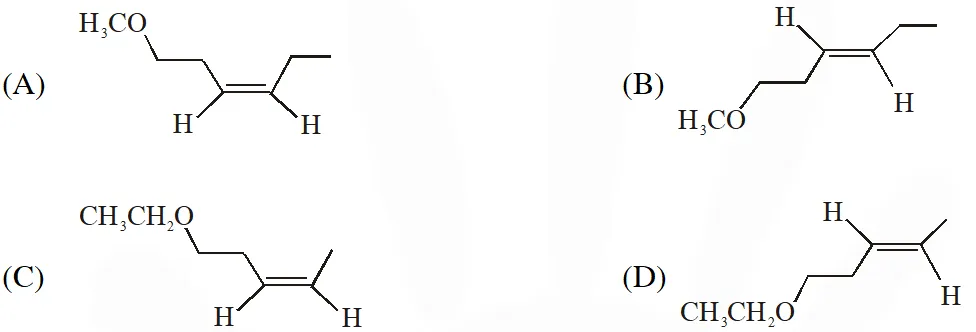 [IIT-2014]
[IIT-2014]
Ans. (A)
Q. The correct statement with respect to prodcut Y is –
(A) It gives a positive Tollens test and is a functional isomer of X
(B) It gives a positive Tollens test and is a geometrical isomer of X
(C) It gives a positive Iodoform test and is a functional isomer of X
(D) It gives a positive Iodoform test and is a geometrical isomer of X
[IIT-2014]
Ans. (C )
Q. Compound(s) that on hydrogenation produce(s) optically inactive compound(s) is (are) –
 [IIT-2015]
[IIT-2015]
 [IIT-2015]
[IIT-2015]
Ans. (B,D)

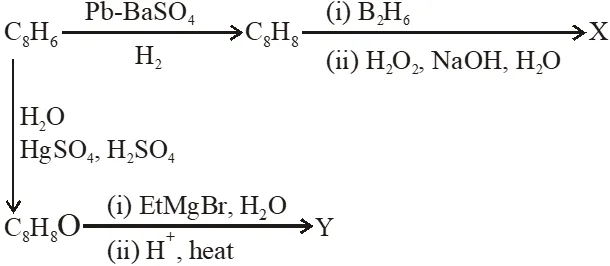
Q. Compound X is :
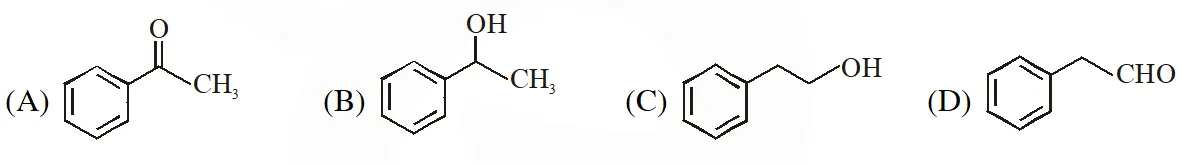 [IIT-2015]
[IIT-2015]
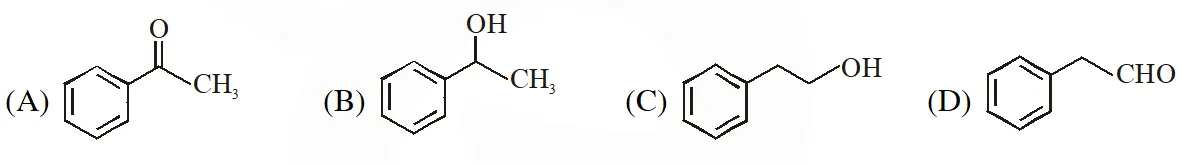 [IIT-2015]
[IIT-2015]
Ans. (C)
Q. The major compound Y is :
 [IIT-2015]
[IIT-2015]
 [IIT-2015]
[IIT-2015]
Ans. (D)
