Hydrocarbons - JEE Main Previous Year Questions with Solutions
JEE Main Previous Year Papers Questions of Chemistry With Solutions are available at eSaral.
Simulator
Previous Years AIEEE/JEE Mains Questions
Q. The main product of the following reaction is
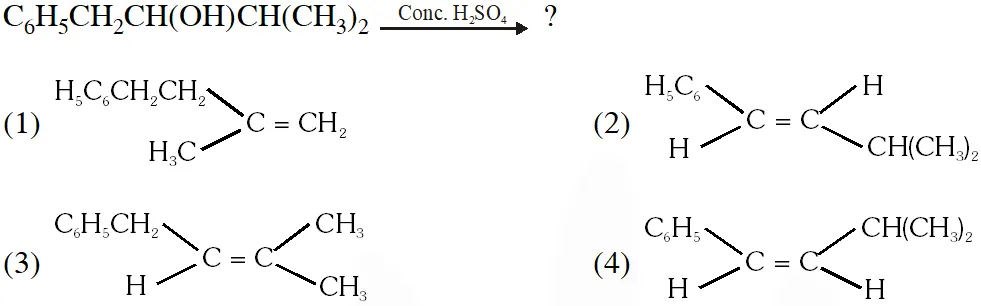 [AIEEE-2010]
[AIEEE-2010]
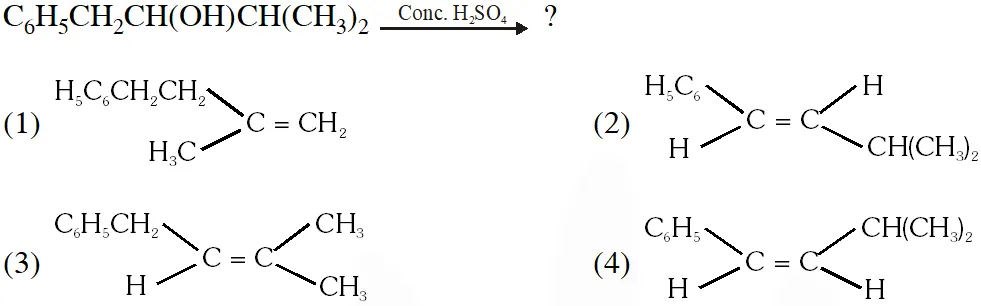 [AIEEE-2010]
[AIEEE-2010]
Ans. (2)
Q. One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of 44 u. The alkene is :-
(1) Ethene
(2) Propene
(3) 1-Butene
(4) 2-Butene
[AIEEE-2010]
Ans. (4)
Q. Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of :-
(1) an isopropyl group
(2) an acetylenic triple bond
(3) two ethylenic double bonds
(4) a vinyl group
[AIEEE-2011]
Ans. (4)
Q. Ozonolysis of an organic compound 'A' produces acetone and propionaldehyde in equimolar mixture. Identify 'A' from the following compounds :-
(1) 2-Methyl-1-pentene
(2) 1-Pentene
(3) 2-Pentene
(4) 2-Methyl-2-pentene
[AIEEE-2011]
Ans. (4)
Q. 2–Hexyne gives trans–2–Hexene on treatment with :-
 [AIEEE-2012]
[AIEEE-2012]
Ans. (3)
Q. In the given transformation, which of the following is the most appropriate reagent ?
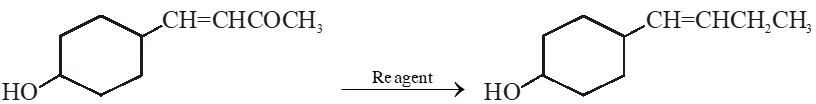
 [AIEEE-2012]
[AIEEE-2012]

Ans. (2)
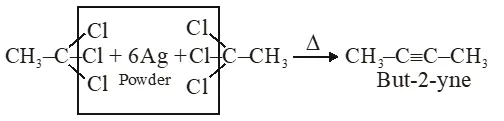
Q. The major organic compound formed by the reaction of 1, 1, 1–trichloroethane with silver powder is :-
(1) 2-Butyne
(2) 2-Butene
(3) Acetylen
(4) Ethene
[Jee-Main offline 2013]
Ans. (1)

Q. The number and type of bonds in $\mathrm{C}_{2}^{2-}$ ion in $\mathrm{CaC}_{2}$ are:
(1) Two $\sigma$ bonds and one $\pi$ - bond
(2) Two $\sigma$ bonds and two $\pi$ - bonds
(3) One $\sigma$ bond and two $\pi$ - bonds
(4) One $\sigma$ bond and one $\pi-$ bond
[Jee-Main offline 2014]
Ans. (3)
$\mathrm{Ca}^{+2}[\mathrm{C} \equiv \mathrm{C}]^{-2}$
Q. In the hydroboration - oxidation reaction of propene with diborane, $\mathrm{H}_{2} \mathrm{O}_{2}$ and NaOH, the organic compound formed is :
(1) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{OH}$
( 2)$\left(\mathrm{CH}_{3}\right)_{3} \mathrm{COH}$
(3) $\mathrm{CH}_{3} \mathrm{CHOHCH}_{3}$
(4) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{OH}$
[Jee-Main offline 2014]
Ans. (1)
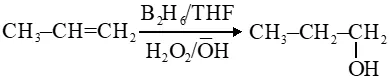
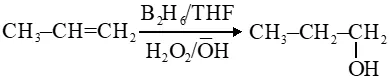
Q. Which one of the following class of compounds is obtained by polymerization of acetylene ?
(1) Poly-ene
(2) Poly-yne
(3) Poly-amide
(4) Poly-ester
[Jee-Main offline 2014]
Ans. (2)
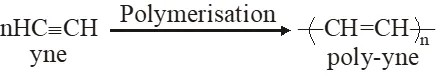
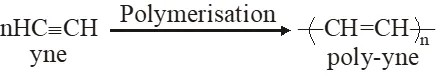
Q. The gas liberated by the electrolysis of Dipotassium succinate solution is :
(1) Ethyne
(2) Ethene
(3) Propene
(4) Ethane
[Jee-Main offline 2014]
Ans. (2)
Q. The reagent needed for converting
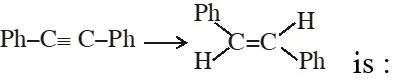 (1) $\mathrm{H}_{2} /$ lindlar Cat.
(2) Cat. Hydrogenatio
(1) $\mathrm{H}_{2} /$ lindlar Cat.
(2) Cat. Hydrogenatio
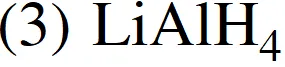 (4) LiNH $_{3}$
[Jee-Main offline 2014]
(4) LiNH $_{3}$
[Jee-Main offline 2014]
Ans. (4)
Q. 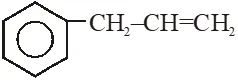 on mercuration- demercuration produces the major product :-
on mercuration- demercuration produces the major product :-
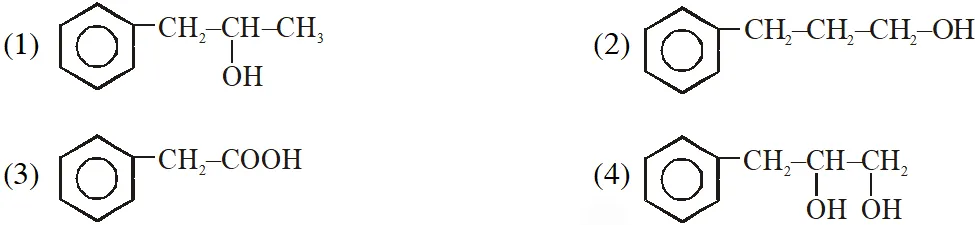 [Jee-Main offline 2014]
[Jee-Main offline 2014]
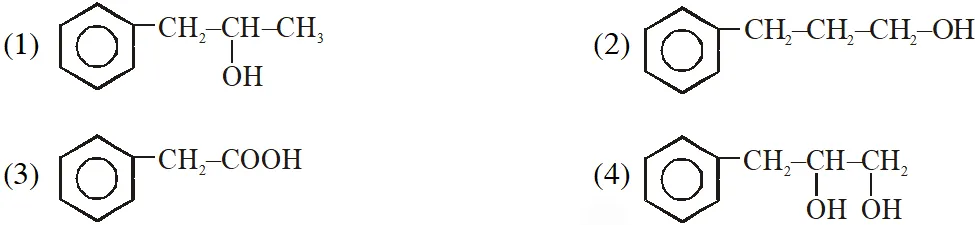 [Jee-Main offline 2014]
[Jee-Main offline 2014]
Ans. (1)
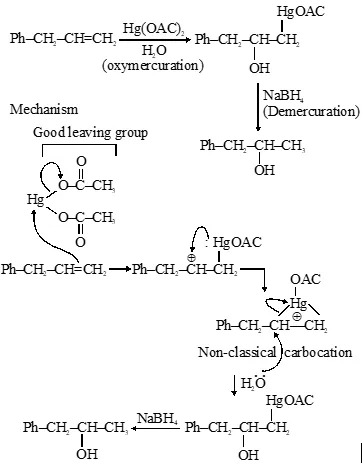 Rearrangement of carbocation formed is not possible due to formatiion of cyclic non-classical carbocation.
Rearrangement of carbocation formed is not possible due to formatiion of cyclic non-classical carbocation.
 Rearrangement of carbocation formed is not possible due to formatiion of cyclic non-classical carbocation.
Rearrangement of carbocation formed is not possible due to formatiion of cyclic non-classical carbocation.
Q. The major product obtained in the photo catalysed bromination of 2-methylbutane is :-
(1) 2-bromo-2-methylbutane
(2) 2-bromo-3-methylbutane
(3) l-bromo-2-methylbutane
(4) l-bromo-3-methylbutane
[Jee-Main offline 2014]
Ans. (1)
Q. In the presence of peroxide, HC and HI do not give anti-Markownikoff s addition to alkenes because
(1) All the steps are exothermic in HCl and HI
(2) One of the steps is endothermic in HCl and HI
(3) HCl is oxidizing and the HI is reducing
(4) Both HCl and HI are strong acids
[Jee-Main offline 2014]
Ans. (2)
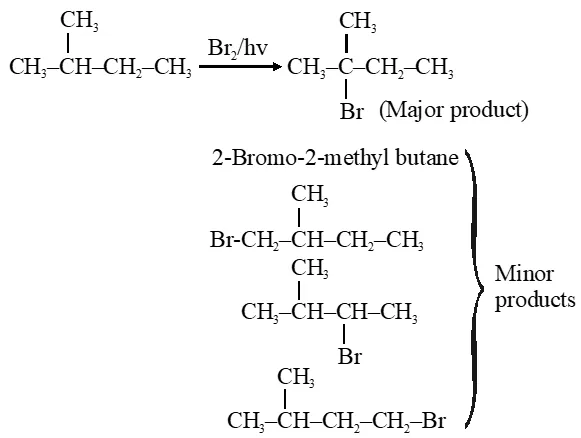 relectivity ratio for bromination is
$1^{\circ}: 2^{\circ}: 3^{\circ}: 1: 82: 1600$
Hence $3^{\circ}$ product will be major product.
relectivity ratio for bromination is
$1^{\circ}: 2^{\circ}: 3^{\circ}: 1: 82: 1600$
Hence $3^{\circ}$ product will be major product.
 relectivity ratio for bromination is
$1^{\circ}: 2^{\circ}: 3^{\circ}: 1: 82: 1600$
Hence $3^{\circ}$ product will be major product.
relectivity ratio for bromination is
$1^{\circ}: 2^{\circ}: 3^{\circ}: 1: 82: 1600$
Hence $3^{\circ}$ product will be major product.
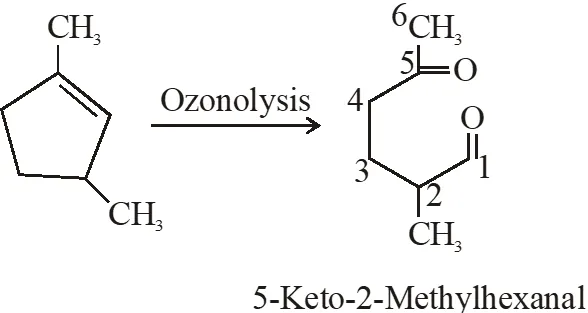
Q. Which compound would give 5-keto-2-methyl hexanal upon ozonlysis?
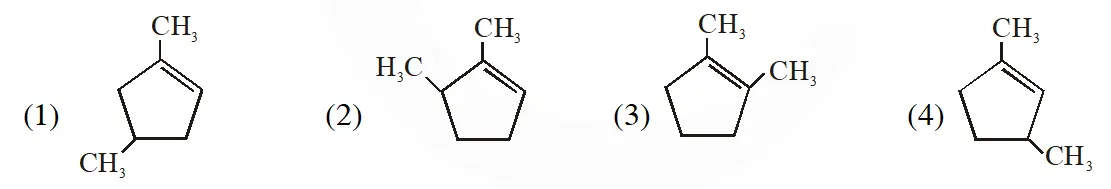 [Jee-Main offline 2015]
[Jee-Main offline 2015]
 [Jee-Main offline 2015]
[Jee-Main offline 2015]
Ans. (4)

Q. The trans-alkenes are formed by the reduction of alkynes with :
(1) $\mathrm{NaBH}_{4}$
(2) Na/liq. NH $_{3}$
(3) Sn–HCl
(4) $\mathrm{H}_{2}-\mathrm{Pd} / \mathrm{C}, \mathrm{BaSO}_{4}$
[Jee-Main offline 2018]
Ans. (2)
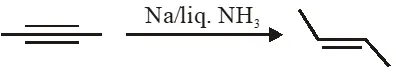 Birch reduction is anti addition. So trans alkene will be produced.
Birch reduction is anti addition. So trans alkene will be produced.
Comments
Aditya H
Jan. 22, 2021, 3:08 p.m.
we can't download e saral app in laptops....so it would be good if u concentrate here as much as you concentrate on your app
radheshysm bhagat
Dec. 24, 2020, 8:06 a.m.
give some hard questions these questions are to easy
by the way good questions ti solve
Dar suhail
Aug. 21, 2020, 11 a.m.
thanks so much for providing these important question..... please add some more questions so that we can clean our concepts.....again thanks ...
