Ionic Equilibrium- JEE Advanced Previous Year Questions with Solutions
JEE Advanced Previous Year Questions of Chemistry with Solutions are available at eSaral. Practicing JEE Advanced Previous Year Papers Questions of Chemistry will help the JEE aspirants in realizing the question pattern as well as help in analyzing weak & strong areas.
Simulator
Previous Years JEE Advance Questions
Q. The dissociation constant of a substituted benzoic acid at $25^{\circ} \mathrm{C}$ is $1.0 \times 10^{-4}$ . The pH of a 0.01 M solution of its sodium salt is
[JEE 2009]
Ans. 8
$\mathrm{K}_{\mathrm{h}}=\frac{\mathrm{K}_{\mathrm{w}}}{\mathrm{K}_{\mathrm{a}}}=\frac{10^{-14}}{10^{-4}}=10^{-10}$
$\mathrm{h}=\sqrt{\frac{\mathrm{K}_{\mathrm{h}}}{\mathrm{C}_{0}}}=\sqrt{\frac{10^{-10}}{10^{-2}}}=10^{-4}$
$\mathrm{pH}=\frac{1}{2}[\mathrm{pKw}+\mathrm{pKa}+\log \mathrm{Co}]$
$\mathrm{pH}=\frac{1}{2}\left[14+4+\log 10^{-2}\right]$
$\mathrm{pH}=\frac{1}{2}[16]=8$
Q. Aqueous solutions of $\mathrm{HNO}_{3}, \mathrm{KOH}, \mathrm{CH}_{3} \mathrm{COOH}$ and $\mathrm{CH}_{3} \mathrm{COONa}$ of identical concentrations are provided. The pair(s) of solutions which form a buffer upon mixing is(are)
(A) $\mathrm{HNO}_{3}$ and $\mathrm{CH}_{3} \mathrm{COOH}$
(B) $\mathrm{KOH}$ and $\mathrm{CH}_{3} \mathrm{COONa}$
(C) $\mathrm{HNO}_{3}$ and $\mathrm{CH}_{3} \mathrm{COONa}$
(D) $\mathrm{CH}_{3} \mathrm{COOH}$ and $\mathrm{CH}_{3} \mathrm{COONa}$
Ans. (C,D)
Q. In 1 L saturated solution of AgCl $\left[\mathrm{K}_{\mathrm{sp}}(\mathrm{AgCl})=1.6 \times 10^{-10}\right], 0.1 \mathrm{mol}$ of $\mathrm{CuCl}$
$\left[\mathrm{K}_{\mathrm{sp}}(\mathrm{CuCl})=1.0 \times 10^{-6}\right]$ is added.
The resultant concentration of $\mathrm{Ag}^{+}$ in the solution is 1.6 × $10^{-x}$. The value of ‘x’ is.
[JEE -2011]
Ans. 7
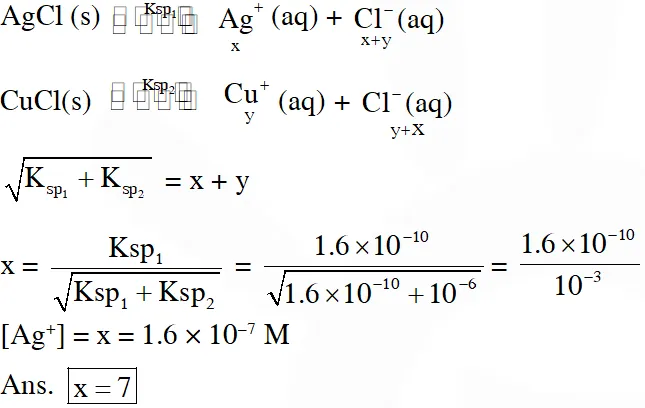
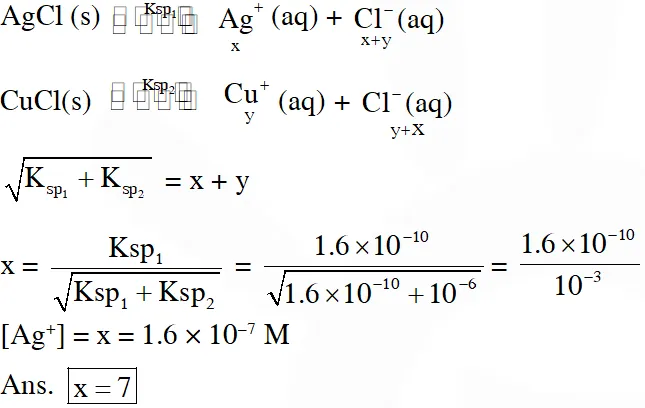
Q. The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is $1 / 100^{\text {th }}$ of that of a strong acid (HX, 1M), at $25^{\circ} \mathrm{C}$. The $\mathrm{K}_{\mathrm{a}}$ of HA is
(A) $1 \times 10^{-4}$
(B) $1 \times 10^{-5}$
(C) $1 \times 10^{-6}$
(D) $1 \times 10^{-3}$
[JEE 2013]
Ans. (A)
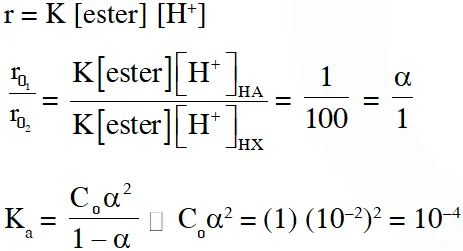
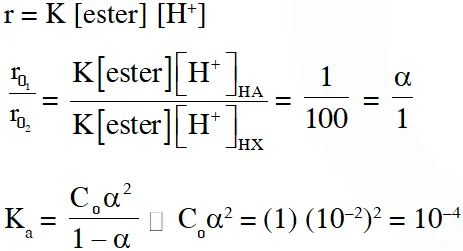
Q. The $\mathrm{K}_{\mathrm{sp}}$ of $\mathrm{Ag}_{2} \mathrm{CrO}_{4}$ is $1.1 \times 10^{-12}$ at 298 K. The solubility (in mol/L) of $\mathrm{Ag}_{2} \mathrm{CrO}_{4}$ in a $0.1 \mathrm{M} \mathrm{AgNO}_{3}$solution is
(A) $1.1 \times 10^{-11}$
(B) $1.1 \times 10^{-10}$
(C) $1.1 \times 10^{-12}$
(D) $1.1 \times 10^{-9}$
[JEE 2013]
Ans. (B)
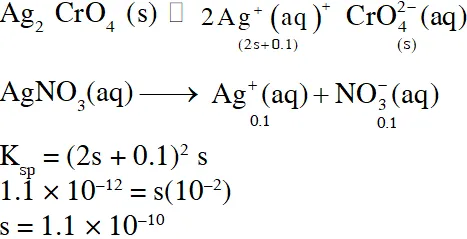
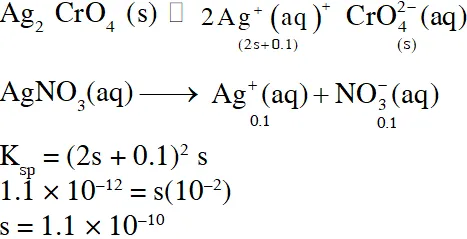
Q. Dilution process of different aqueous solutions; with water, are given in LIST-I. The
effects of dilution of the solutions on $\left[\mathrm{H}^{+}\right]$ are given in LIST II.
(Note : Degree of dissociation () of weak acid and weak base is << 1; degree of hydrolysis of salt <<1; $\left[\mathrm{H}^{+}\right]$ represents the concentration of $\mathrm{H}^{+}$ ions)
 Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
$(\mathrm{A}) \mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 2 ; \mathrm{R} \rightarrow 3 ; \mathrm{S} \rightarrow 1$
(B) $\mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 3 ; \mathrm{R} \rightarrow 2 ; \mathrm{S} \rightarrow 3$
(C) $\mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 4 ; \mathrm{R} \rightarrow 5 ; \mathrm{S} \rightarrow 3$
$(\mathrm{D}) \mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 5 ; \mathrm{R} \rightarrow 4 ; \mathrm{S} \rightarrow 1$
[JEE- Adv. - 2018]
Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
$(\mathrm{A}) \mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 2 ; \mathrm{R} \rightarrow 3 ; \mathrm{S} \rightarrow 1$
(B) $\mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 3 ; \mathrm{R} \rightarrow 2 ; \mathrm{S} \rightarrow 3$
(C) $\mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 4 ; \mathrm{R} \rightarrow 5 ; \mathrm{S} \rightarrow 3$
$(\mathrm{D}) \mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 5 ; \mathrm{R} \rightarrow 4 ; \mathrm{S} \rightarrow 1$
[JEE- Adv. - 2018]
 Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
$(\mathrm{A}) \mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 2 ; \mathrm{R} \rightarrow 3 ; \mathrm{S} \rightarrow 1$
(B) $\mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 3 ; \mathrm{R} \rightarrow 2 ; \mathrm{S} \rightarrow 3$
(C) $\mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 4 ; \mathrm{R} \rightarrow 5 ; \mathrm{S} \rightarrow 3$
$(\mathrm{D}) \mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 5 ; \mathrm{R} \rightarrow 4 ; \mathrm{S} \rightarrow 1$
[JEE- Adv. - 2018]
Match each process given in LIST-I with one or more effect(s) in LIST-II. The correct option is
$(\mathrm{A}) \mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 2 ; \mathrm{R} \rightarrow 3 ; \mathrm{S} \rightarrow 1$
(B) $\mathrm{P} \rightarrow 4 ; \mathrm{Q} \rightarrow 3 ; \mathrm{R} \rightarrow 2 ; \mathrm{S} \rightarrow 3$
(C) $\mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 4 ; \mathrm{R} \rightarrow 5 ; \mathrm{S} \rightarrow 3$
$(\mathrm{D}) \mathrm{P} \rightarrow 1 ; \mathrm{Q} \rightarrow 5 ; \mathrm{R} \rightarrow 4 ; \mathrm{S} \rightarrow 1$
[JEE- Adv. - 2018]
Ans. (D)
 correct match : Q-5
correct match : Q-5

 correct match : Q-5
correct match : Q-5

