NCERT Solutions for Class 9 Science chapter 1 - Matter in our Surrounding - eSaral
Hey, are you a class 9 student and looking for ways to download NCERT Solutions for Class 9 Science chapter 1 "Matter in our Surrounding"? If yes. Then you are at the right place.
In this article, we have listed NCERT solutions for class 9 Science Chemistry Chapter 1 in PDF that is prepared by Kota’s top expert Faculties by keeping Simplicity in mind.
If you want to learn and understand class 9 Science chapter 1 "Matter in our Surrounding" in an easy way then you can use these solutions PDF.
NCERT Solutions helps students to prepare for important concepts. Class 9 Science solutions provide detailed explanations of all the questions that students can use to clear their doubts instantly.
If you want to score high in your class 9 Science Exam then it is very important for you to have a good knowledge of all the important topics, so to learn and practice those topics you can use NCERT Solutions.
In this article, we have listed NCERT Solutions for Class 9 Science chapter 1 that you can download to start your preparations anytime.
India's Best Exam Preparation for Class 9th - Download Now
So, without wasting more time Let’s start
Download The PDF of NCERT Solutions for Class 9 Science Chapter 1 "Matter in our Surroundings"








Download The PDF of NCERT Solutions for Class 9 Science Chapter 1 "Matter in our Surroundings"
India's Best Exam Preparation for Class 9th - Download Now
Important topics of class 9 Science Chapter 1
- Matter in our Surroundings
- Physical Nature of Matter
- Characteristics of Particles of Matter
- Evaporation
- States of Matter
- Can Matter change its state?
So, that’s all from this article. I hope you enjoyed this post. If you found this article helpful then please share it with other students.
All Questions of Chapter 1
Once you Complete the chapter 1 then you can revise it by solving following questions.
Q1. Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold-drink, smell of perfum
Q2. Give reasons for the following observation:
The smell of hot sizzling food reaches you several meters away, but to get the smell from cold
food you have to go close.
Q3. A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Q4. What are the characteristics of the particles of matter?
Part 2
Q1. The mass per unit volume of a substance is called density.
(density = mass/volume).
Arrange the following in order of increasing density: air, exhaust from chimneys, honey, water,
chalk, cotton and iron.
Q2. (a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: rigidity, compressibility, fluidity, fliling a gas container, shape, kinetic energy and density.
Q3. Give reasons :
(a) A gas fills completely the vessel in which it is kept.
(b) A gas exerts pressure on the walls of the container.
(c) A wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood we need a karate expert.
Q4. Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why.
India's Best Exam Preparation for Class 9th - Download Now
Part 3
Q1. Convert the following temperature to Celsius scale :
(a) 300 K
(b) 573 K
Q2. What is the physical state of water at :
(a) 250°C
(b) 100°C
Q3. For any substance, why does the temperature remain constant during the change of state?
Q4. Suggest a method to liquefy atmospheric gases?
Part 4
Q1. Why does a desert cooler cool better on a hot dry day?
Q2. How does the water kept in an earthen pot (matka) become cool during summer?
Q3. Why does our palm feel cold when we put some acetone or petrol or perfume on it?
Q4. Why are we able to sip hot tea or milk faster from a saucer rather than a cup?
Q5. What type of clothes should we wear in summer?
Exercise
1. Convert the following temperature to the Celsius scale -
(i) 293 K
(ii) 470 K
2. Convert the following temperature to the kelvin scale.
(i) 25°C
(ii) 373°C
3. (a) Naphthalene balls disappear with time without leaving any solid why?
(b) We can get the smell of perfume sitting several meters away.
4. Arrange the following substances in the increasing order of forces of attraction between the particles – water, sugar and oxygen.
5 What is the physical state of water at
(a) 25°C
(b) 0°C
(c) 100°C
6. Give two reasons to justify.
(a) Water at room temperature is a liquid.
(b) An iron almirah is solid at room temperature.
7. Ice at 273 K is more effective in cooling, than water at the same temperature, why?
8. What produces more severe burns, boiling water or steam?
9. Name A, B, C, D, E and F in the following diagram showing change in its state. 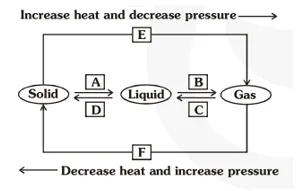
Also Read,
India's Best Exam Preparation for Class 9th - Download Now
Science Class 9 Chapterwise Revision notes
NCERT Class 9 Science Book PDF
Class 9 Science Syllabus
NCERT Class 9 Science Exemplar
If you have any Confusion related to NCERT Solutions for Class 9 Science chapter 1 then feel free to ask in the comments section down below.
To watch Free Learning Videos on Class 9 by Kota’s top Faculties Install the eSaral App
