Properties of Diamagnetic Materials - Magnetism & Matters | Class 12 Physics Notes
Here we will study about the Paramagnetic and Diamagnetic Materials. Properties of Diamagnetic Materials & Paramagnetic Substances also discussed in this article.
Diamagnetic Materials
- The substances which when placed in a magnetising field get feebly magnetised in a direction opposite to magnetising field are called diamagnetic.
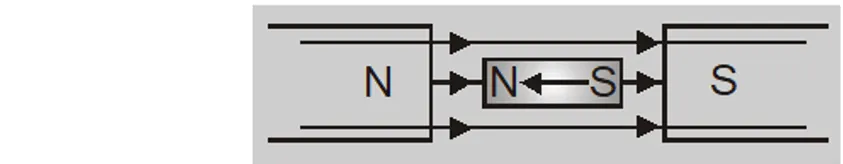
- The substances are weakly repelled by the field so in a non uniform field these have a tendency to move from strong to weak field.
- A diamagnetic rod sets itself perpendicular to field because field is strongest at poles.
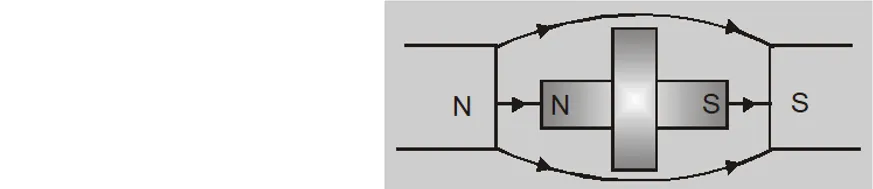
- A diamagnetic liquid in a U-tube depresses in the limb which is between the poles of magnet.

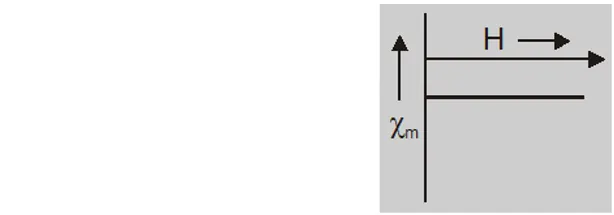
- Intensity of magnetisation I is very small, negative and proportional to magnetising field.

- Magnetic susceptibility $\chi_{\mathrm{m}}=\frac{\mathrm{I}}{\mathrm{H}}$ is small and negative $\left(\approx 10^{-5}\right)$
- The relative permeability $\mu_{\mathrm{r}}=\frac{\mu}{\mu_{0}}$ is slightly less than unity.
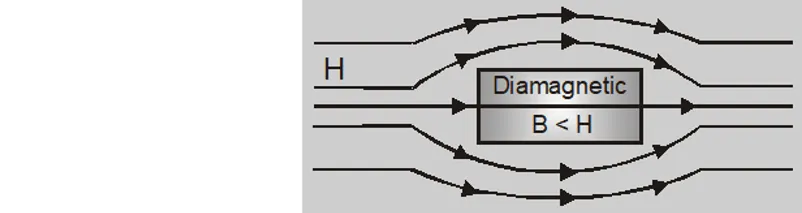
- The field inside the material $\mathrm{B}$ is less than magnetising field H. They have a tendency to expel lines of force.
- Magnetic dipole moment (M) is small and opposite to magnetising field H.
- Diamagnetic substances do not obey Curie's law and show no transition
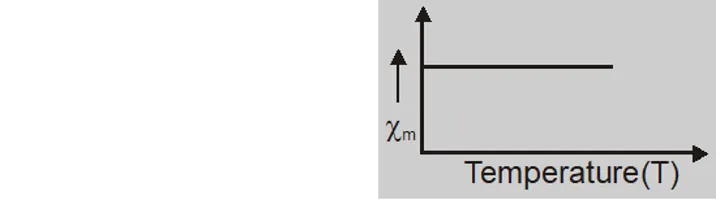
- at Curie temperature $X_{m}$ is indepedent of temperature.
- The origin of diamagnetism is the induced dipole moment due to change in orbital motion of electrons in atoms by applied field.
- Imp. Diamagnetism is present in all materials and is independent of temperature. As it is weak it is often masked by para and ferromagnetic effects.
- The atoms do not have any permanent dipole moment i.e. paired spin.
- Diamagnetism is exhibited by solids, liquids and gases.
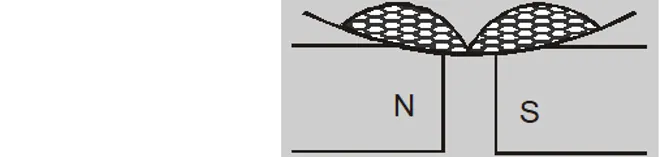
- If a diamagnetic liquid is placed in a watch glass placed. on two pole pieces which are quite close to each other then liquid accumulates at sides and shows depression in the middle, where field is strongest.
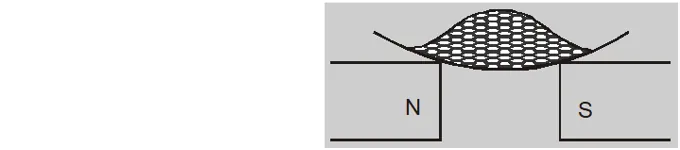
- If a diamagnetic liquid is placed in a watch glass placed on two pole pieces which are sufficiently apart then liquid accumulates in the middle where field is weakest.
About eSaral At eSaral we are offering a complete platform for IIT-JEE & NEET preparation. The main mission behind eSaral is to provide education to each and every student in India by eliminating the Geographic and Economic factors, as a nation’s progress and development depends on the availability of quality education to each and every one. With the blend of education & technology, eSaral team made the learning personalized & adaptive for everyone.For free video lectures and complete study material, Download eSaral APP.
