(a) What are hydrocarbons ? Give examples.
(b) Give the structural differences between saturated and unsaturated hydrocarbons with two examples of each.
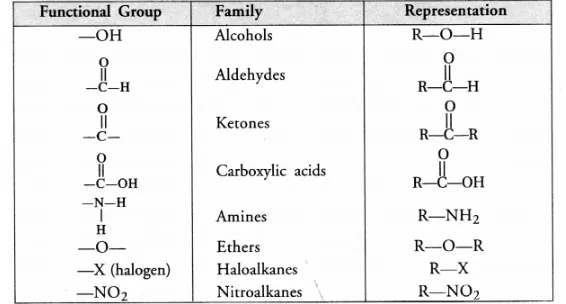
(c) What is a functional group ? Give examples of four different functional groups.
(a) Hydrocarbons are the organic compounds containing only carbon and hydrogen atoms as their constituents. These may be alkanes, alkenes and
alkynes.
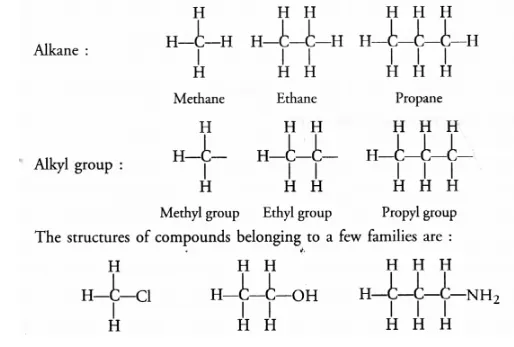
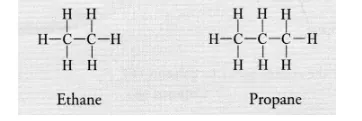
(b) Saturated hydrocarbons or alkanes contain either C—C or C—H bonds in their molecules. These are represented by the general formula
CnH2n+2 For example,
Unsaturated hydrocarbons contain either atleast one >C = C< bond or triple —C ≡ C— bond in their molecules. These may be either alkenes or
alkynes in nature. The general formula of alkenes is CnH2n while that of alkynes is CnH2n-2 for example,
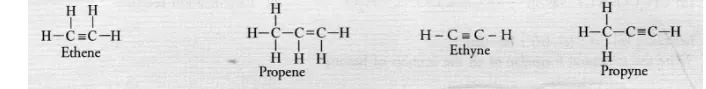
(c) For the definition of functional group and example,