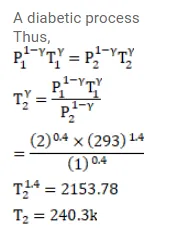Air (γ = 1.4) is pumped at 2atm pressure in a motor tyre at 20°c.
Question:
Air $(\gamma=1.4)$ is pumped at 2 atm pressure in a motor tyre at $20^{\circ} \mathrm{C}$. If the tyre suddenly bursts, what would be the temperature of the air coming out of the tyre. Neglect any mixing with the atmospheric air.
Solution: