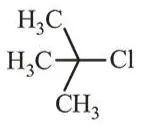
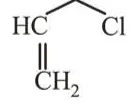
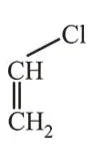
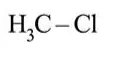Question:
Among the following compounds, which one has the shortest $\mathrm{C}-\mathrm{Cl}$ bond?
Correct Option: , 4
Solution:
Due to conjugation of lonepair of $\mathrm{Cl}$ with $\pi$ bond, partial double bond character decreases bond length that's why compound (d) has shortest $\mathrm{C}-\mathrm{Cl}$ bond length.