Question:
Consider the given plot of enthalpy of the following reaction between $\mathrm{A}$ and $\mathrm{B}$.
$\mathrm{A}+\mathrm{B} \rightarrow \mathrm{C}+\mathrm{D}$
Identify the incorrect statement.
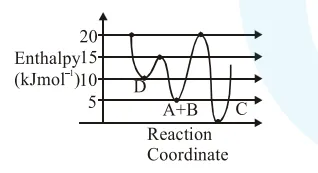
Correct Option: , 4
Solution:
$\mathrm{A}+\mathrm{B} \rightarrow \mathrm{C}+\mathrm{D}$
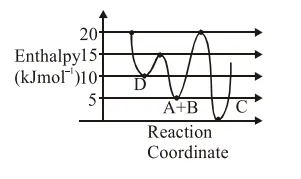
Activation enthalpy for $\mathrm{C}=20-5=15 \mathrm{~kJ} / \mathrm{mol}$
Activation enthalpy for $\mathrm{D}=15-5=10 \mathrm{~kJ} / \mathrm{mol}$
