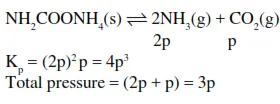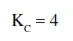In reaction
Question:
In reaction $\mathrm{A}+2 \mathrm{~B} \rightleftharpoons 2 \mathrm{C}+\mathrm{D}$, initial concentration of $\mathrm{B}$ was $1.5$ times of $|\mathrm{A}|$, but at equilibrium the concentrations of $\mathrm{A}$ and $\mathrm{B}$ became equal. The equilibrium constant for the reaction is :
Correct Option: 1,
Solution: