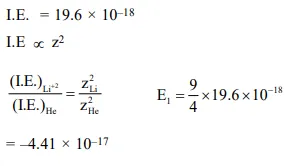Ionisation energy of
Question:
Ionisation energy of $\mathrm{He}^{+}$is $19.6 \times 10^{-18} \mathrm{~J}_{\text {atom }^{-1}}$. The energy of the first stationary state $(\mathrm{n}=1)$
Correct Option: , 3
Solution: