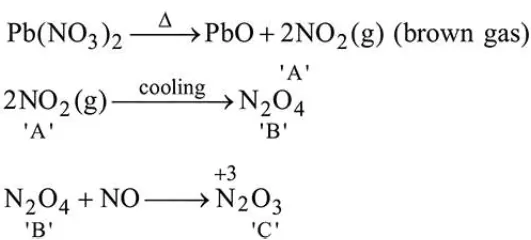
Question:
On heating, lead (II) nitrate gives a brown gas (A). The gas (A) on cooling changes to a colourless solid/liquid (B). (B) on heating with NO changes to a blue solid (C). The oxidation number of nitrogen in solid (C) is :
Correct Option: , 3
Solution: