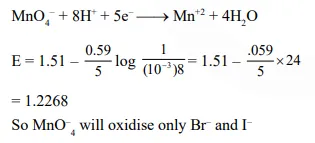Question: At $298 \mathrm{~K}$, the standard reduction potentials are $1.51 \mathrm{~V}$ for $\mathrm{MnO}_{4}^{-} \mid \mathrm{Mn}^{2+}, 1.36 \mathrm{~V}$ for $\operatorname{oxidize}\left(\frac{\mathrm{RT}}{\mathrm{F}}=0.059 \mathrm{~V}\right):-$
$\mathrm{Cl}^{-}$and $\mathrm{Br}^{-}$
$\mathrm{Cl}^{-}, \mathrm{Br}^{-}$and $\mathrm{I}^{-}$
$\mathrm{Br}^{-}$and $\mathrm{I}^{-}$
I' $^{-}$only
Correct Option:
Solution: