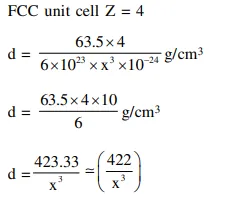Question:
At $100^{\circ} \mathrm{C}$, copper $(\mathrm{Cu})$ has $\mathrm{FCC}$ unit cell structure with cell edge length of $x \AA$. What is the approximate density of $\mathrm{Cu}\left(\right.$ in $\mathrm{g} \mathrm{cm}^{-3}$ ) at this temperature ?
[Atomic Mass of Cu $=63.55 \mathrm{u}$ ]
Correct Option: , 4
Solution: