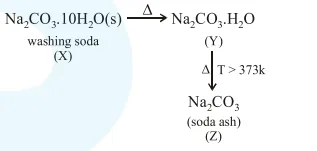
Question: A hydrated solid $X$ on heating initially gives a monohydrated compound $Y$. Y upon heating above $373 \mathrm{~K}$ leads to an anhydrous white powder $\mathrm{Z} . \mathrm{X}$ and $\mathrm{Z}$, respectively, are:
Washing soda and soda ash.
Washing soda and dead burnt plaster.
Baking soda and dead burnt plaster.
Baking soda and soda ash.
Correct Option: 1
Solution: